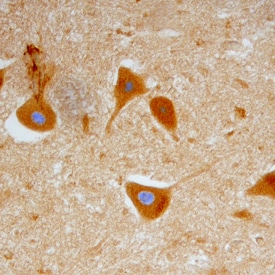
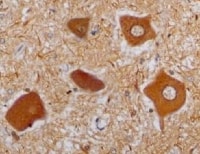
Glial cell line-derived growth factor (GDNF), neurturin (NTN), artemin and persephin are distant members of the TGF-beta superfamily. They function as neurotrophic factors for a variety of neuronal populations in the central and peripheral nervous systems. The bioactivities of GDNF and NTN are mediated through a receptor complex composed of the non ligand-binding signaling subunit (c-Ret receptor tyrosine kinase) and either of two ligand binding subunits [GDNF receptor alpha -1 (GFR alpha -1) or GFR alpha -2]. GFR alpha -1 and -2 are members of a family of at least four cysteine-rich glycosyl-phosphatidylinositol (GPI)-linked cell surface proteins that share conserved placements of many of their cysteine residues. Binding of GDNF to membrane-associated GFR alpha -1 or GFR alpha -2 initiates the association with and activation of the Ret tyrosine kinase. Soluble GFR alpha s released enzymatically from the cell surface-associated protein with phosphatidylinositol phospholipase C, as well as recombinantly produced soluble GFR alpha -1, can also bind with high-affinity to GDNF and trigger the activation of Ret tyrosine kinase.
Human GFR alpha -1 cDNA encodes a 465 amino acid (aa) residue protein with an N-terminal 24 aa residue hydrophobic signal peptide. Like other GPI-linked proteins, human GFR alpha -1 has a C-terminal hydrophobic region which is preceded by a three aa residue (ASS) GPI-binding site. Human GFR alpha -1 shares 93% aa identity with rat GFR alpha -1. The expression of the various GFR alpha s are differentially regulated in the central and peripheral nervous system, suggesting complementary roles for the GFR alpha s in mediating the activities of the GDNF family of neurotrophic factors.