Recombinant Human TNF-alpha, Biotinylated Protein
R&D Systems | Catalog # BT210

Loading...
Key Product Details
- R&D Systems E. coli-derived Recombinant Human TNF-alpha, Biotinylated Protein (BT210)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
E. coli
Accession Number
Structure / Form
Homotrimer.
Biotinylated protein via amines
Conjugate
Biotin
Applications
Bioactivity
Loading...
Product Specifications
Source
E. coli-derived human TNF-alpha protein
Val77-Leu233, with and without an N-terminal Met
Val77-Leu233, with and without an N-terminal Met
Purity
>95%, by SDS-PAGE visualized with Silver Staining and quantitative densitometry by Coomassie® Blue Staining.
Endotoxin Level
<0.10 EU per 1 μg of the protein by the LAL method.
N-terminal Sequence Analysis
Val77
Predicted Molecular Mass
17 kDa (unlabeled)
SDS-PAGE
17 kDa, reducing conditions
Activity
Measured in a cytotoxicity assay using L‑929 mouse fibroblast cells in the presence of the metabolic inhibitor actinomycin D. Matthews, N. and M.L. Neale (1987) in Lymphokines and Interferons, A Practical Approach. Clemens, M.J. et al. (eds): IRL Press. 221.
The ED50 for this effect is 25-100 pg/mL.
The ED50 for this effect is 25-100 pg/mL.
Scientific Data Images for Recombinant Human TNF-alpha, Biotinylated Protein
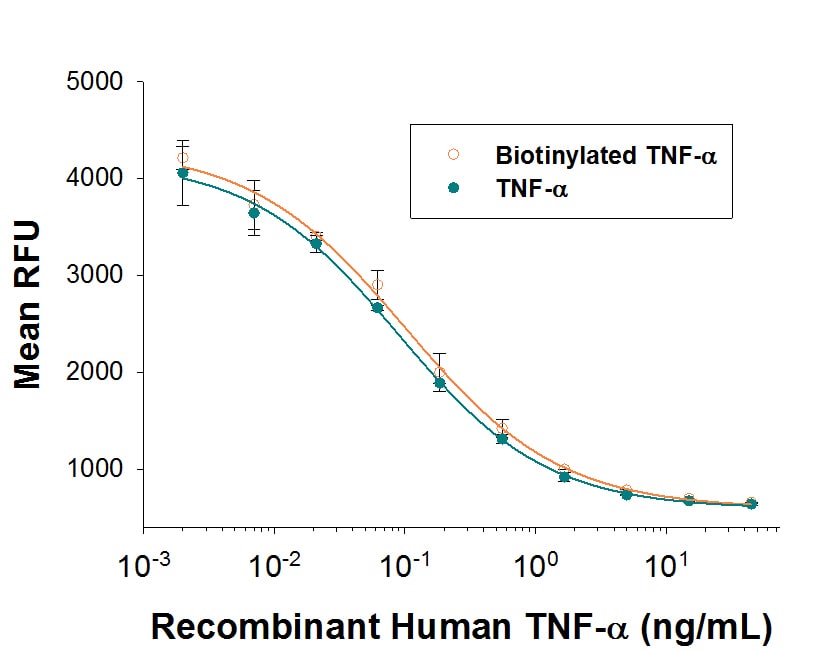
Recombinant Human TNF-alpha, Biotinylated Protein Bioactivity
Both Biotinylated Recombinant Human TNF-a (Catalog # BT210) and unlabeled Recombinant Human TNF-a (Catalog # 210-TA) promotes cytotoxicity in L-929 mouse fibroblast cells in the presence of the metabolic inhibitor actinomycin D. The ED50 for this effect is 25-100 pg/mL. The similarity in activity highlights that the biotinylated protein is fully functional.Formulation, Preparation, and Storage
BT210
| Formulation | Lyophilized from a 0.2 μm filtered solution in PBS with BSA as a carrier protein. |
| Reconstitution | Reconstitute at 100 μg/mL in sterile PBS. |
| Shipping | The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Calculators
Background: TNF-alpha
References
- Zelova, H. and J. Hosek (2013) Inflamm. Res. 62:641.
- Juhasz, K. et al. (2013) Expert Rev. Clin. Immunol. 9:335.
- Pennica, D. et al. (1984) Nature 312:724.
- Tang, P. et al. (1996) Biochemistry 35:8216.
- Perez, C. et al. (1990) Cell 63:251.
- Black, R.A. et al. (1997) Nature 385:729.
- Moss, M.L. et al. (1997) Nature 385:733.
- Gearing, A.J.H. et al. (1994) Nature 370:555.
- Schall, T.J. et al. (1990) Cell 61:361.
- Loetscher, H. et al. (1990) Cell 61:351.
- Dembic, Z. et al. (1990) Cytokine 2:231.
- Smith, C.A. et al. (1990) Science 248:1019.
- Loetscher, H. et al. (1991) J. Biol. Chem. 266:18324.
- Pinckard, J.K. et al. (1997) J. Biol. Chem. 272:10784.
- Engelmann, H. et al. (1990) J. Biol. Chem. 265:1531.
Long Name
Tumor Necrosis Factor alpha
Alternate Names
Cachetin, DIF, TNF, TNF-A, TNFA, TNFalpha, TNFG1F, TNFSF1A, TNFSF2
Entrez Gene IDs
Gene Symbol
TNF
UniProt
Additional TNF-alpha Products
Product Documents for Recombinant Human TNF-alpha, Biotinylated Protein
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Human TNF-alpha, Biotinylated Protein
For research use only
Related Research Areas
Citations for Recombinant Human TNF-alpha, Biotinylated Protein
Customer Reviews for Recombinant Human TNF-alpha, Biotinylated Protein
There are currently no reviews for this product. Be the first to review Recombinant Human TNF-alpha, Biotinylated Protein and earn rewards!
Have you used Recombinant Human TNF-alpha, Biotinylated Protein?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Loading...
Associated Pathways
 Apoptosis Signaling Pathway
Apoptosis Signaling Pathway
 Dendritic Cell Lineage Development Pathways
Dendritic Cell Lineage Development Pathways
 Hematopoietic Stem Cell Differentiation Pathways & Lineage-specific Markers
Hematopoietic Stem Cell Differentiation Pathways & Lineage-specific Markers
 IL-2 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
IL-2 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types

IL-15 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
 Innate Lymphoid Cell Differentiation Pathways
Innate Lymphoid Cell Differentiation Pathways
 mTOR Signaling Pathway
mTOR Signaling Pathway
 NOD-like Receptor Signaling Pathways
NOD-like Receptor Signaling Pathways
 Th1 Differentiation Pathway
Th1 Differentiation Pathway
 TNF Superfamily Pathway: Human Ligand-Receptor Interactions & their Associated Functions
TNF Superfamily Pathway: Human Ligand-Receptor Interactions & their Associated Functions
 Toll-Like Receptor Signaling Pathways
Toll-Like Receptor Signaling Pathways

 Innate Lymphoid Cell Differentiation Pathways
Innate Lymphoid Cell Differentiation Pathways
 mTOR Signaling Pathway
mTOR Signaling Pathway
 NOD-like Receptor Signaling Pathways
NOD-like Receptor Signaling Pathways
 Th1 Differentiation Pathway
Th1 Differentiation Pathway
 TNF Superfamily Pathway: Human Ligand-Receptor Interactions & their Associated Functions
TNF Superfamily Pathway: Human Ligand-Receptor Interactions & their Associated Functions
 Toll-Like Receptor Signaling Pathways
Toll-Like Receptor Signaling Pathways

