Human IL-10 Quantikine HS ELISA Kit
R&D Systems | Catalog # HS100C


Key Product Details
Assay Length
Sample Type & Volume Required Per Well
Sensitivity
Assay Range
Product Summary for Human IL-10 Quantikine HS ELISA Kit
Product Specifications
Measurement
Detection Method
Conjugate
Species
Specificity
Cross-reactivity
Interference
精度
Intra-Assay Precision (Precision within an assay) Three samples of known concentration were tested on one plate to assess intra-assay precision.
Inter-Assay Precision (Precision between assays) Three samples of known concentration were tested in separate assays to assess inter-assay precision.
Citrate Plasma, EDTA Plasma, Serum
| Intra-Assay Precision | Inter-Assay Precision | |||||
|---|---|---|---|---|---|---|
| Sample | 1 | 2 | 3 | 1 | 2 | 3 |
| n | 20 | 20 | 20 | 35 | 35 | 35 |
| Mean (pg/mL) | 2.36 | 10.7 | 21.0 | 1.68 | 10.4 | 20.9 |
| Standard Deviation | 0.22 | 0.62 | 0.97 | 0.22 | 0.81 | 1.78 |
| CV% | 9.4 | 5.8 | 4.6 | 12.8 | 7.8 | 8.5 |
Recovery for Human IL-10 Quantikine HS ELISA Kit
The recovery of IL-10 spiked to three different levels throughout the range of the assay in various matrices was evaluated.
| Sample Type | Average % Recovery | Range % |
|---|---|---|
| Citrate Plasma (n=4) | 93 | 81-106 |
| EDTA Plasma (n=4) | 90 | 84-99 |
| Serum (n=4) | 100 | 94-108 |
Linearity
To assess the linearity of the assay, samples were spiked with high concentrations of IL-10 in various matrices and diluted with Calibrator Diluent to produce samples with values within the dynamic range of the assay.

Scientific Data Images for Human IL-10 Quantikine HS ELISA Kit
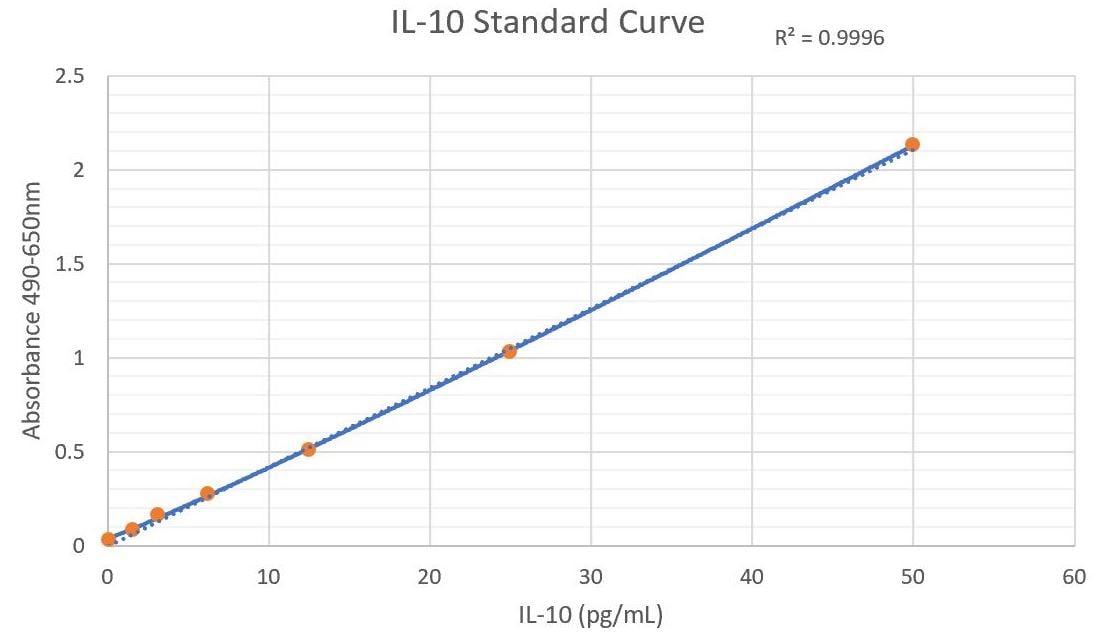
Human IL-10 ELISA Standard Curve
Preparation and Storage
Shipping
Stability & Storage
Background: IL-10
Long Name
Alternate Names
Entrez Gene IDs
Gene Symbol
Additional IL-10 Products
Product Documents for Human IL-10 Quantikine HS ELISA Kit
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human IL-10 Quantikine HS ELISA Kit
For research use only
Related Research Areas
Citations for Human IL-10 Quantikine HS ELISA Kit
Customer Reviews for Human IL-10 Quantikine HS ELISA Kit (6)
Have you used Human IL-10 Quantikine HS ELISA Kit?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
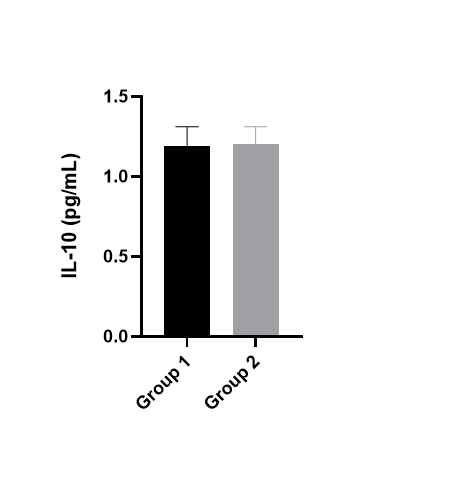
Sample Tested: EDTA PlasmaSpecies: HumanVerified Customer | Posted 05/05/2026Determination of analyte concentrations.
-
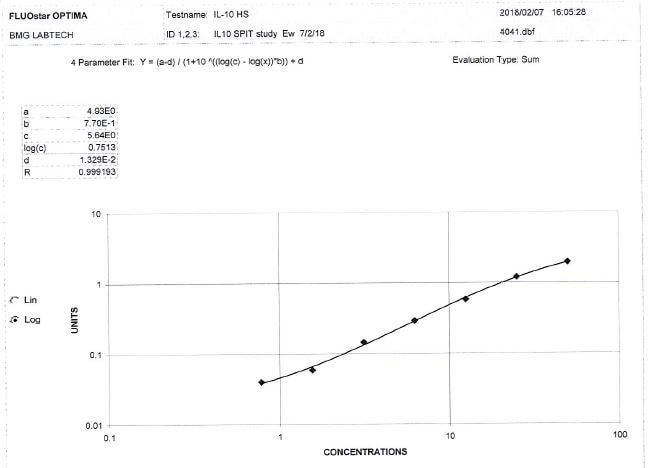
Sample Tested: EDTA Plasma and EDTA PlasmaVerified Customer | Posted 06/27/2023
-
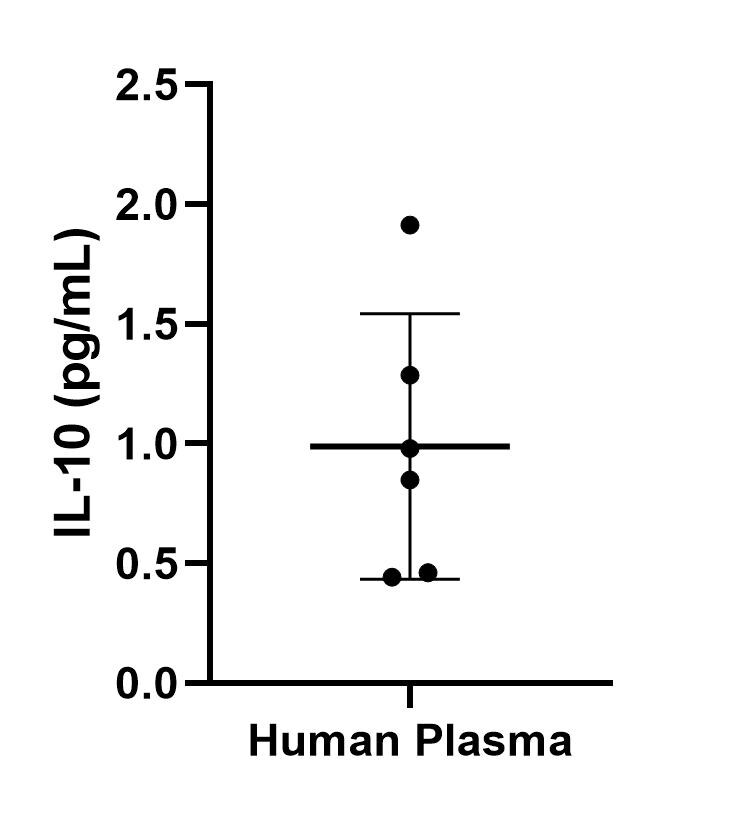
Sample Tested: EDTA PlasmaVerified Customer | Posted 12/06/2021
-
Sample Tested: PlasmaVerified Customer | Posted 02/28/2020Easy to use, clear instructions, good results
-
Sample Tested: PlasmaVerified Customer | Posted 02/27/2020
-
Sample Tested: EDTA PlasmaVerified Customer | Posted 01/18/2018
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- ELISA Sample Preparation & Collection Guide
- ELISA Troubleshooting Guide
- How to Run an R&D Systems DuoSet ELISA
- How to Run an R&D Systems Quantikine ELISA
- How to Run an R&D Systems Quantikine™ QuicKit™ ELISA
- Quantikine HS ELISA Kit Assay Principle, Alkaline Phosphatase
- Quantikine HS ELISA Kit Principle, Streptavidin-HRP Polymer
- Sandwich ELISA (Colorimetric) – Biotin/Streptavidin Detection Protocol
- Sandwich ELISA (Colorimetric) – Direct Detection Protocol
- Troubleshooting Guide: ELISA
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Associated Pathways
 Hematopoietic Stem Cell Differentiation Pathways & Lineage-specific Markers
Hematopoietic Stem Cell Differentiation Pathways & Lineage-specific Markers
 IL-2 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
IL-2 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
 Innate Lymphoid Cell Differentiation Pathways
Innate Lymphoid Cell Differentiation Pathways
 Jak/STAT Signaling Pathway
Jak/STAT Signaling Pathway