Bretazenil
Tocris Bioscience | Catalog # 3568

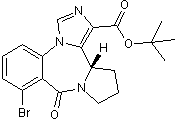
Key Product Details
Description
Alternative Names
Product Description
Bretazenil is a partial agonist at the GABAA benzodiazepine site (EC50 = 10 nM at α1β1γ2 receptors). Displays anticonvulsive activity in vivo.
Product Specifications for Bretazenil
Molecular Weight
Formula
Storage
Purity
Chemical Name
CAS Number
PubChem ID
InChI Key
SMILES
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Solubility
| Solvent | Max Conc. mg/mL | Max Conc. mM | |
|---|---|---|---|
| Solubility | |||
| DMSO | 41.83 | 100 | |
| Ethanol | 41.83 | 100 |
Preparing Stock Solutions for Bretazenil
The following data is based on the product molecular weight 418.28.
Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which all affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 1 mM | 2.39 mL | 11.95 mL | 23.91 mL |
| 5 mM | 0.48 mL | 2.39 mL | 4.78 mL |
| 10 mM | 0.24 mL | 1.20 mL | 2.39 mL |
| 50 mM | 0.05 mL | 0.24 mL | 0.48 mL |
Calculators
Background References
References are publications that support the biological activity of the product.
- Munro Comparison of the novel subtype-selective GABAA receptor-positive allosteric modulator NS11394 [3'-[5-(1-hydroxy-1-methyl-ethyl)-benzoimidazol-1-yl]-biphenyl-2-carbonitrile] with D.pam, zolpidem, bretazenil, and gaboxadol in rat models of i J.Pharmacol.Exp.Ther. 2008 PMID: 18791060
- Puia Molecular mechanisms of the partial allosteric modulatory effects of bretazenil at γ-aminobutyric acid type A receptor. Proc.Natl.Acad.Sci. USA Neurobiology
- Rundfeldt Anticonvulsant tolerance and withdrawal characteristics of benzodiazepine receptor ligands in different seizure models in mice. Comparison of D.pam, bretazenil and abecarnil. J.Pharmacol.Exp.Ther. 1995 PMID: 7473156
Product Documents for Bretazenil
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for Bretazenil
For research use only
Related Research Areas
Citations for Bretazenil
Customer Reviews for Bretazenil
There are currently no reviews for this product. Be the first to review Bretazenil and earn rewards!
Have you used Bretazenil?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
