Calcein AM
Tocris Bioscience | Catalog # 5119

Key Product Details
Description
Wavelength
Product Description
Key information: Calcein AM is a cell permeable non-fluorescent compound, that becomes green fluorescent once hydrolyzed in live cells.Used for: cell tracing, monitoring cell viability, chemotaxis, cell adhesion and multidrug resistance.
Application: fluorescent microscopy and flow cytometry.
Properties and Photophysical Data: in live cells, non-fluorescent Calcein AM is hydrolyzed by intracellular esterases into green-fluorescent calcein, which is retained in the cytoplasm. Excitation and emission maxima (λ) are 495 nm and 515 nm, respectively.
It is recommended to prepare stock solutions of Calcein AM in DMSO.
Optical Data

| Emission Color | Green |
| λabs | 495 nm |
| λem | 515 nm |
| Cell Permeable | Yes |
| Application | Flow Cytometry, Fluorescent Microscopy |
Spectra Viewer
Plan Your Experiments
Use our spectra viewer to interactively plan your experiments, assessing multiplexing options. View the excitation and emission spectra for our fluorescent dye range and other commonly used dyes.
Spectra Viewer
Product Specifications for Calcein AM
Molecular Weight
Formula
Storage
Purity
Chemical Name
CAS Number
PubChem ID
InChI Key
SMILES
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Calculators
Background References
References are publications that support the biological activity of the product. See our Citations tab to view 20 publications citing the usage of this product.
- De Gendt The use of calcein acetomethylester (AM)-labelled polymorphonuclear cells in a polycarbonate filter chemotaxis assay. Clin.Chim.Acta. 1996 PMID: 8737602
- Lazarowski Direct demonstration of mechanically induced release of cellular UTP and its implication for uridine nucleotide receptor activation. J.Biol.Chem. 1997 PMID: 9305892
- Kuehn Prostaglandin E2 activates and utilizes mTORC2 as a central signaling locus for the regulation of mast cell chemotaxis and mediator release. J.Biol.Chem. 2011 PMID: 20980255
- Bakos Membrane topology and glycosylation of the human multidrug resistance-associated protein. J.Biol.Chem. 1996 PMID: 8647833
Product Documents for Calcein AM
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for Calcein AM
For research use only
Citations for Calcein AM
Customer Reviews for Calcein AM (4)
Have you used Calcein AM?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Species: RatAssay Type: In VitroCell Line/Tissue: Spinal neuronsVerified Customer | Posted 09/11/2025To measure toxicity in rat spinal cord neurons
-
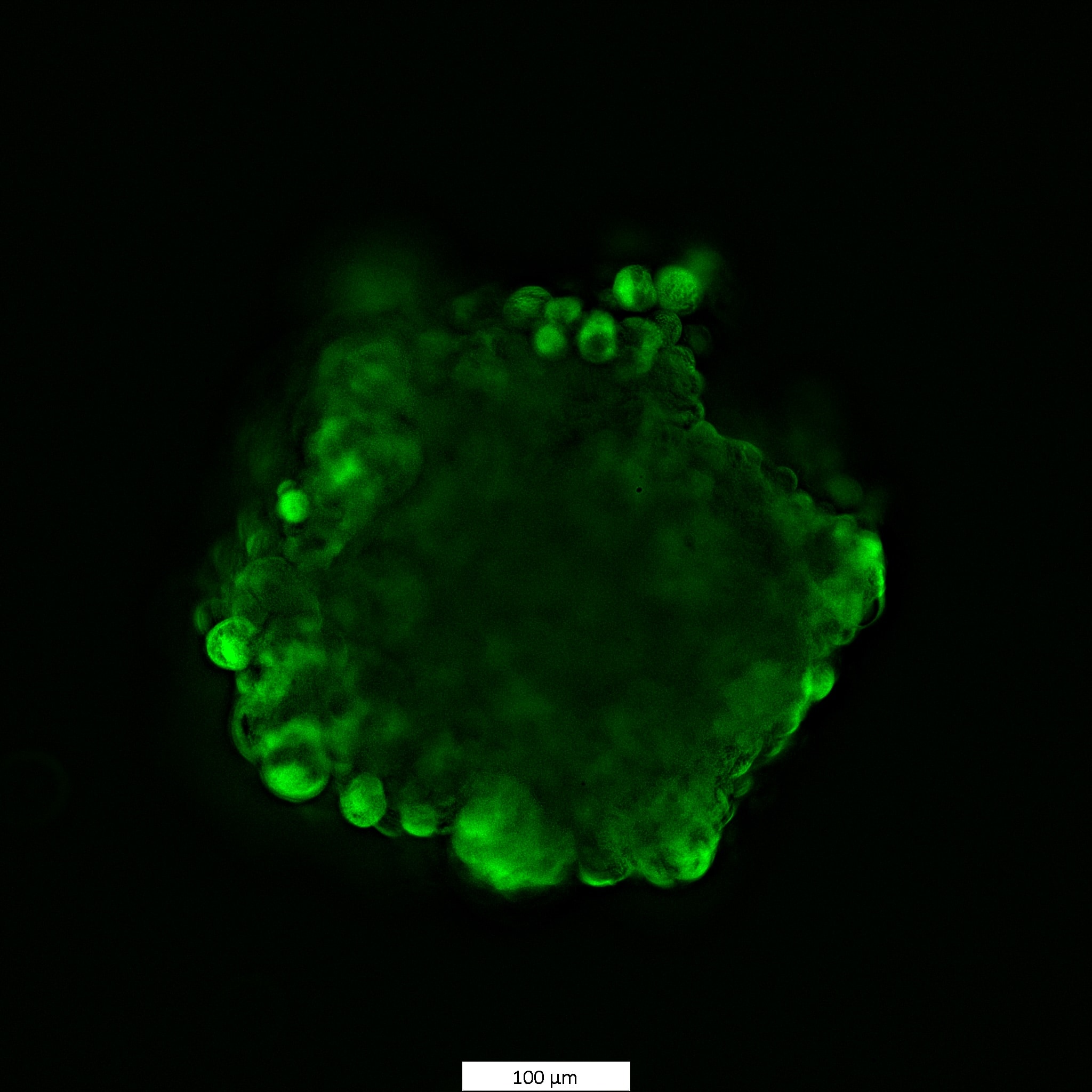
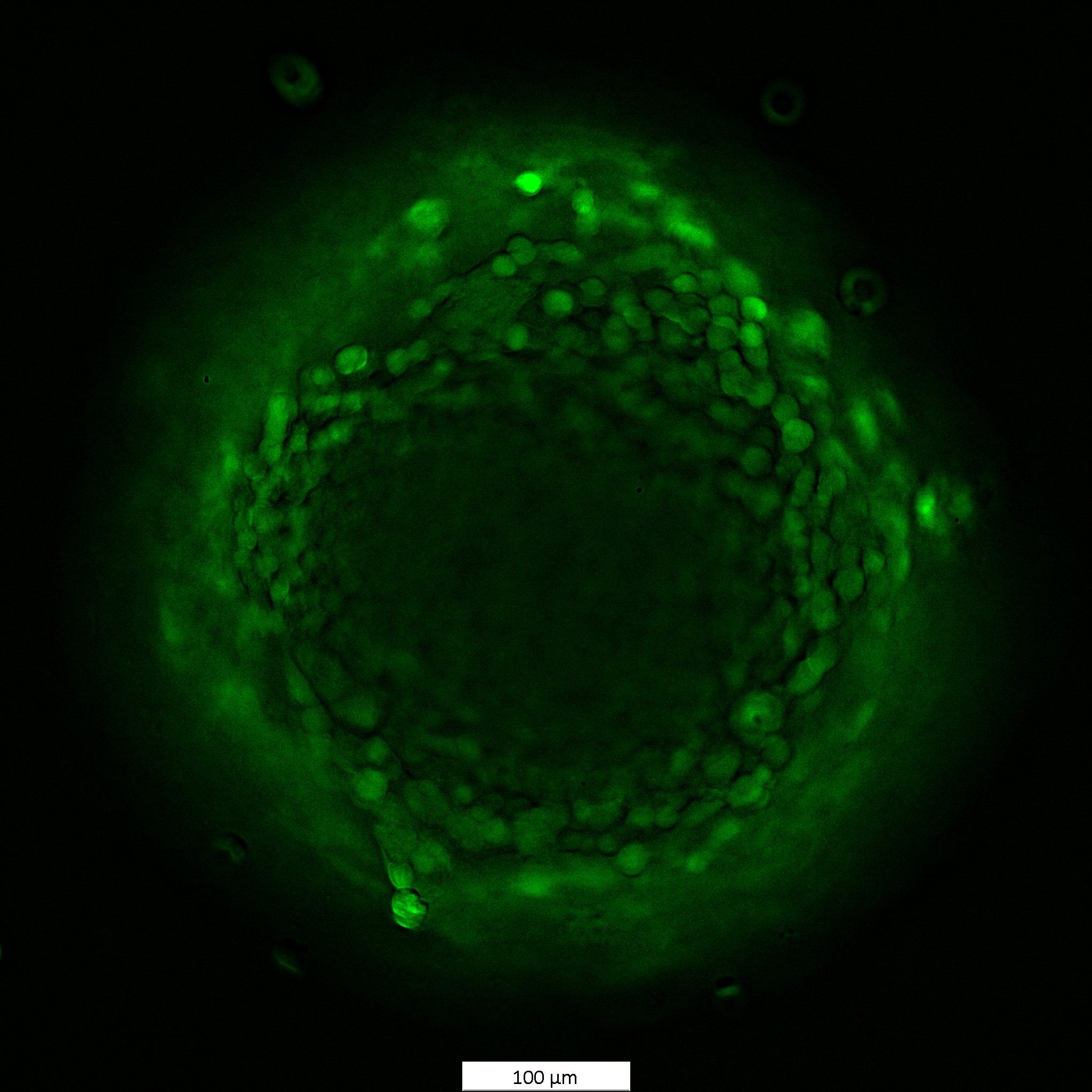
Species: HumanAssay Type: In VitroCell Line/Tissue: MCF7Verified Customer | Posted 11/26/2024MCF7 cells spheroids were stained with 1 uM Calcein AM for 1.5h
-
Species: HumanAssay Type: In VitroCell Line/Tissue: HCT116Verified Customer | Posted 11/26/2024Viability staining of HCT116 spheroids 1 uM concentration
-
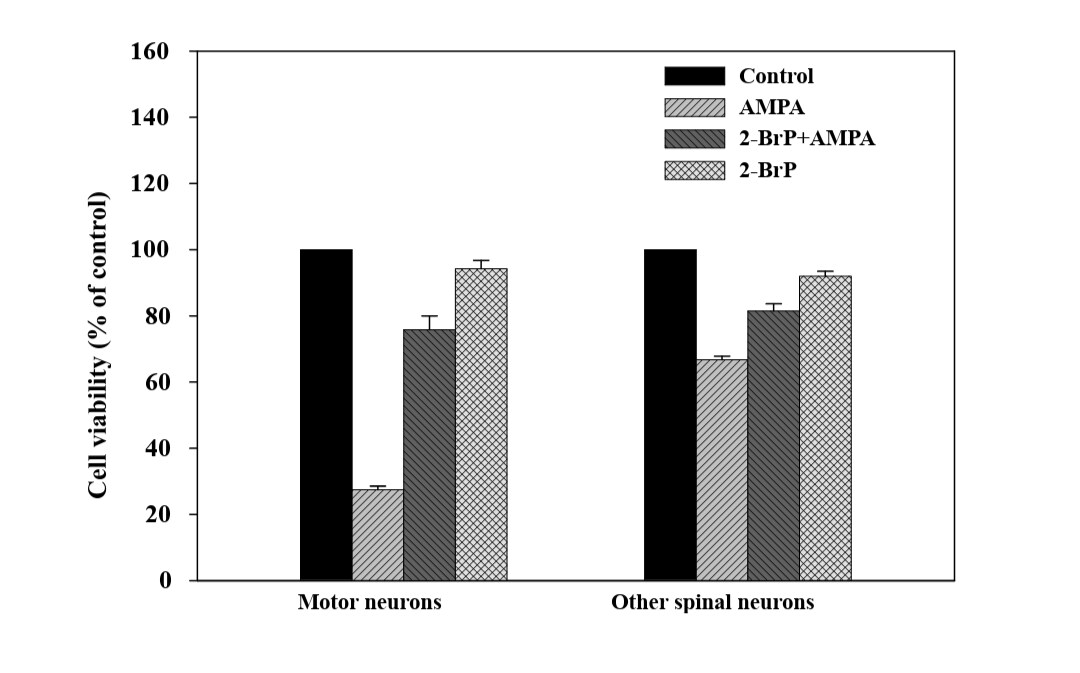
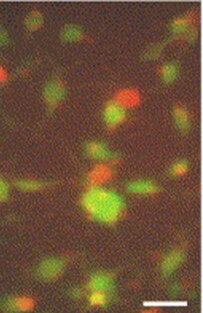
Species: HumanAssay Type: In VitroCell Line/Tissue: motor neuronsVerified Customer | Posted 10/24/2018Neurons were treated with 50 uM glutamate for 1 hour. To assess the neuronal viability, neurons were incubated with calcein-AM (2 uM), and ethidium homodimer (4 uM) in HEPES buffer for 45-60 min at 37 degrees, rinsed with HEPES 4-5 times and examined under an epifluorescence microscope. This assay clearly distinguished live cells stained positive for calcein (green) from the dead cells stained positive for ethidium homodimer (red).For analyzing cellular toxicity in neurons
There are no reviews that match your criteria.