Bicalutamide
Tocris Bioscience | Catalog # 3389

Key Product Details
Description
Alternative Names
Product Description
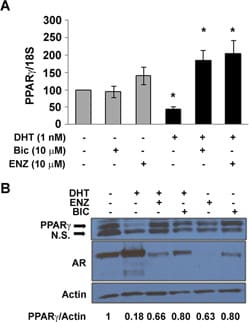
Bicalutamide is an orally active non-steroidal androgen receptor antagonist (IC50 = 190 nM). Displays peripheral selectivity and does not effect serum levels of LH and testosterone. Exhibits potent anticancer activity in vivo.Licensing Information
Sold for research purposes only under agreement from AstraZeneca
Product Specifications for Bicalutamide
Molecular Weight
Formula
Storage
Purity
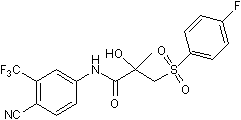
Chemical Name
CAS Number
PubChem ID
InChI Key
SMILES
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Solubility
| Solvent | Max Conc. mg/mL | Max Conc. mM | |
|---|---|---|---|
| Solubility | |||
| DMSO | 43.04 | 100 | |
| Ethanol | 4.3 | 10 |
Preparing Stock Solutions for Bicalutamide
The following data is based on the product molecular weight 430.37.
Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which all affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 1 mM | 2.32 mL | 11.62 mL | 23.24 mL |
| 5 mM | 0.46 mL | 2.32 mL | 4.65 mL |
| 10 mM | 0.23 mL | 1.16 mL | 2.32 mL |
| 50 mM | 0.05 mL | 0.23 mL | 0.46 mL |
Calculators
Background References
References are publications that support the biological activity of the product. See our Citations tab to view 11 publications citing the usage of this product.
- Tucker Nonsteroidal antiandrogens. Synthesis and structure-activity relationships of 3-substituted derivatives of 2-hydroxypropionanilides. J.Med.Chem. 1988 PMID: 3361581
- Furr and Tucker The preclinical development of bicalutamide: pharmacodynamics and mechanism of action. Urology 1996 PMID: 8560673
- Furr ICI 176,334: a novel non-steroidal, peripherally selective antiandrogen. J.Endocrinol. 1987 PMID: 3625091
Product Documents for Bicalutamide
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for Bicalutamide
For research use only
Related Research Areas
Citations for Bicalutamide
Customer Reviews for Bicalutamide (1)
Have you used Bicalutamide?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Species: HumanAssay Type: In VitroCell Line/Tissue: c4-2 cellVerified Customer | Posted 11/03/2019in vitro culture with c4-2 cells to induce AR reduction
There are no reviews that match your criteria.