Devazepide
Tocris Bioscience | Catalog # 2304

Key Product Details
Description
Alternative Names
Product Description
Devazepide is a potent, orally active CCK1 (CCK-A) receptor antagonist that displays appetite-stimulant effects. Blocks the anorectic response to CCK-8 and increases food intake in rats following systemic and i.c.v administration.
Product Specifications for Devazepide
Molecular Weight
Formula
Storage
Purity
Chemical Name
CAS Number
PubChem ID
InChI Key
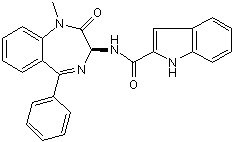
SMILES
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Solubility
| Solvent | Max Conc. mg/mL | Max Conc. mM | |
|---|---|---|---|
| Solubility | |||
| DMSO | 40.85 | 100 | |
| Ethanol | 20.42 | 50 |
Preparing Stock Solutions for Devazepide
The following data is based on the product molecular weight 408.46.
Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which all affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 1 mM | 2.45 mL | 12.24 mL | 24.48 mL |
| 5 mM | 0.49 mL | 2.45 mL | 4.90 mL |
| 10 mM | 0.24 mL | 1.22 mL | 2.45 mL |
| 50 mM | 0.05 mL | 0.24 mL | 0.49 mL |
Calculators
Background References
References are publications that support the biological activity of the product. See our Citations tab to view 45 publications citing the usage of this product.
- Duca MetF. activates a duodenal Ampk-dependent pathway to lower hepatic glucose production in rats. Nat.Med. 2015 PMID: 25849133
- Reidelberger Effects of peripheral CCK receptor blockade on food intake in rats. Am.J.Physiol.Reg.Integr.Comp.Physiol. 2003 PMID: 12738611
- Ritter Increased food intake and CCK receptor antagonists: beyond abdominal vagal afferents. Am.J.Physiol.Reg.Integr.Comp.Physiol. 2004 PMID: 15142854
- Ebenezer Effects of intracerebroventricular administration of the CCK1 receptor antagonist devazepide on food intake in rats. Eur.J.Pharmacol. 2002 PMID: 12007923
Product Documents for Devazepide
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for Devazepide
For research use only
Citations for Devazepide
Customer Reviews for Devazepide (1)
Have you used Devazepide?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
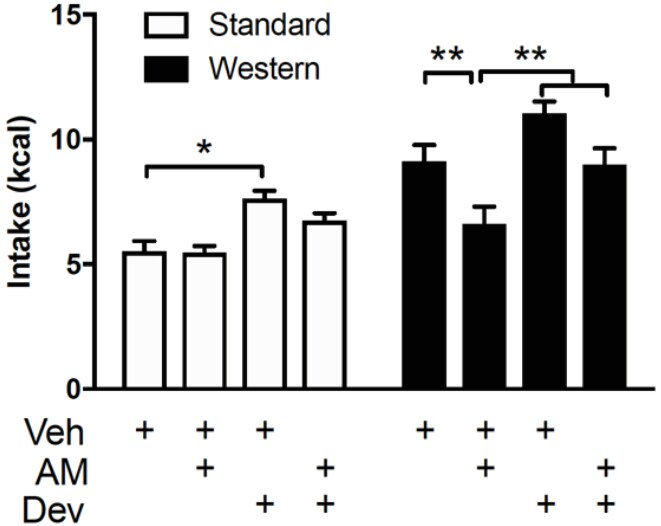
Species: MouseAssay Type: In VivoCell Line/Tissue: CCK-eGFP-positive cellsVerified Customer | Posted 02/10/2021Devazepide was dissolved in vehicle consisting of 7.5% DMSO, 7.5% Tween80, and 85% sterile saline, and warmed in a water bath to ensure solubility.We used low-fat chow fed mice and that is blocked by co-administration with the CCKA receptor antagonist by devazepide treatment. 0.1 mg per kg for devazepide treatment
There are no reviews that match your criteria.