GMP Antibodies
Recombinant unconjugated GMP antibodies for immune cell therapy manufacturing.
GMP Antibodies for T Cell Activation
Add flexibility to your immune cell therapy manufacturing workflow with new GMP-grade (Good Manufacturing Practice) anti-CD3 and anti-CD28 antibodies. Improve the phenotypic profiles of your immune cells by optimizing CD3/CD28 antibody ratios for fine-tuned cell activation. These soluble monoclonal antibodies allow for maximum flexibility to optimize protocols to your specific platform and cell cultures, with high-quality formulations, scalability, detection specificity, low endotoxin levels, and the raw material traceability needed for cGMP reagents.
Available Antibodies | |
|---|---|
GMP Antibodies | Equivalent RUO |
| GMP Anti-CD3 (OKT3) | Anti-CD3 (OKT3) |
| GMP Anti-CD28 (15e8) | Anti-CD28 (15e8) |
Equivalent RUO (research use only) and GMP antibodies simplify the transition to the clinic.
Activation Unlocked
Optimize antigen receptor activation to your platform and cells
Recombinantly Produced
Secure supply chain and easily scalable manufacturing
Reformatted OKT3 and 15e8 Clones
With human backbone to reduce risk associated with non-human FC receptors
Customizable Formats
Flexible fill size and format
Long Expiration Dates
Plan confidently with > 1 year shelf life
Quality Control
ISO 9001, ISO 13485, enhanced QC testing
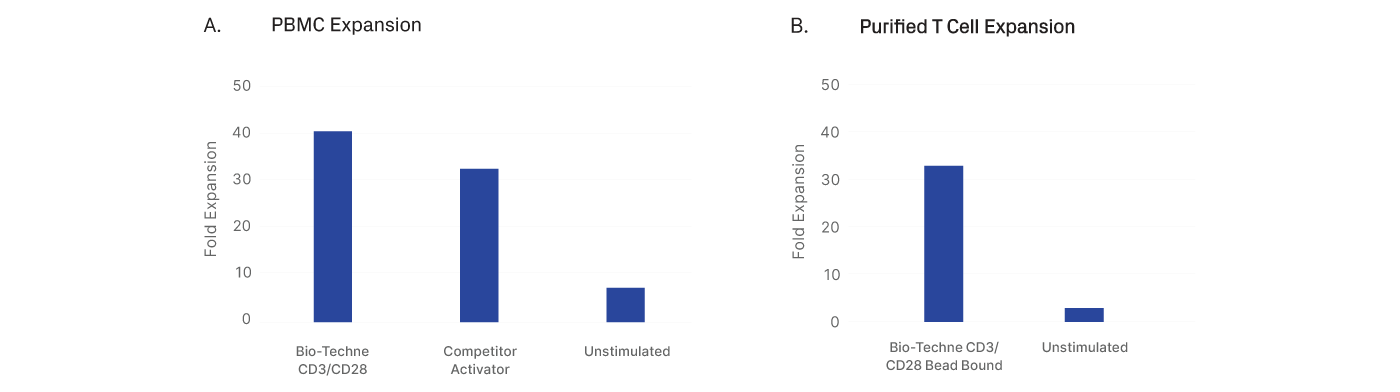
CD3 and CD28 GMP antibodies can be adapted to your protocol regardless of the starting population.
PBMCs from two independent donors were thawed and activated using CD3 and CD28 antibodies. The cells were cultured for 9-days in standard 6-well G-Rex® bioreactors using GMP T Cell Media (Cat. # CCM038-GMP) supplemented with 10 ng/mL of IL-7 (Cat. # BT-007-GMP) and IL-15 (Cat. # BT-015-GMP) cytokines. Each bar in the plots above is the average of two donors. A) PBMCs cultured with Bio-Techne soluble CD3 and CD28 antibodies have higher fold expansion compared to a competitor activator. B) Biotin-labeled CD3 and CD28 antibodies were conjugated with streptavidin magnetic beads and used to activate purified T cells, supporting robust expansion.
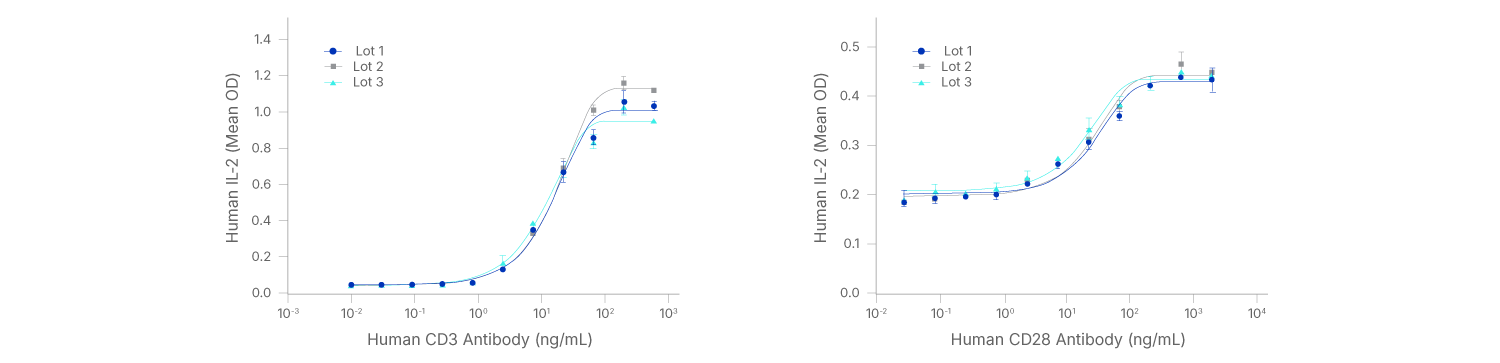
GMP Antibodies Show High Lot-to-Lot Consistency in Functional Assays
GMP CD3 and CD28 Antibodies Perform Similarly Across Multiple Lots.
Human CD3 GMP Antibody (Catalog # MAB11411-GMP) stimulates IL-2 secretion in Jurkat cells (immortalized human T cell line) treated with 5 ng/mL phorbol myristate acetate (PMA), left. The ED50 for this effect is 8.00-80.0 ng/mL. Human CD28 GMP Antibody (Catalog # MAB11412-GMP) enhances IL-2 secretion in Jurkat cells treated with 5 ng/mL PMA and 0.9 µM calcium ionomycin, right. The ED50 for this effect is 5.00-50.0 ng/mL.
Antibody-mediated stimulation occurred in a dose-dependent manner, as measured using the Quantikine Human IL-2 ELISA kit. Data from 3 separate production lots are superimposed in each figure.
T Cell Activation and Expansion Protocols | |
Starting Cells | Antibody Protocol |
T Cells | CD3 Immobilized, CD28 Soluble |
| Bead-Conjugated | |
PBMCs | Soluble and Bead-Conjugated |
GMP Manufacturing Facility
All Bio-Techne GMP antibodies are manufactured recombinantly in a GMP facility, to ensure consistent performance and secure supply. Our dedicated GMP antibody production teams strive to ensure lot-to-lot consistency and minimize supply chain roadblocks that can cause expensive manufacturing process delays.