Flumazenil
Tocris Bioscience | Catalog # 1328

Key Product Details
Description
Alternative Names
Product Description
Flumazenil is a benzodiazepine antagonist, non-selective for α1, α2, α3 or α5-containing GABAA receptors. Centrally active upon systemic administration in vivo. Also inhibits repressor element-1 silencing transcription factor (REST) by targeting degradation.
Product Specifications for Flumazenil
Molecular Weight
Formula
Storage
Purity
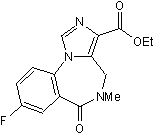
Chemical Name
CAS Number
PubChem ID
InChI Key
SMILES
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Solubility
| Solvent | Max Conc. mg/mL | Max Conc. mM | |
|---|---|---|---|
| Solubility | |||
| DMSO | 25 |
Preparing Stock Solutions for Flumazenil
The following data is based on the product molecular weight 303.29.
Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which all affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 0.25 mM | 13.19 mL | 65.94 mL | 131.89 mL |
| 1.25 mM | 2.64 mL | 13.19 mL | 26.38 mL |
| 2.5 mM | 1.32 mL | 6.59 mL | 13.19 mL |
| 12.5 mM | 0.26 mL | 1.32 mL | 2.64 mL |
Calculators
Background References
References are publications that support the biological activity of the product. See our Citations tab to view 42 publications citing the usage of this product.
- Dobl New insights into the mechanism of action of hypnotics. J.Psychopharmacol. 1999 PMID: 10667451
- Polc Electrophysiological studies on the specific benzodiazepine antagonist Ro 15-1788. Naunyn Schmiedebergs Arch.Pharmacol. 1981 PMID: 7196507
- Atack Regional differences in the inhibition of mouse in vivo [3H]Ro 15-1788 binding reflect selectivity for α1 versus α2 and α3 subunit-containing GABAA receptors. Neuropsychopharmacology 1999 PMID: 10063485
Product Documents for Flumazenil
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for Flumazenil
For research use only
Related Research Areas
Citations for Flumazenil
Customer Reviews for Flumazenil (2)
Have you used Flumazenil?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
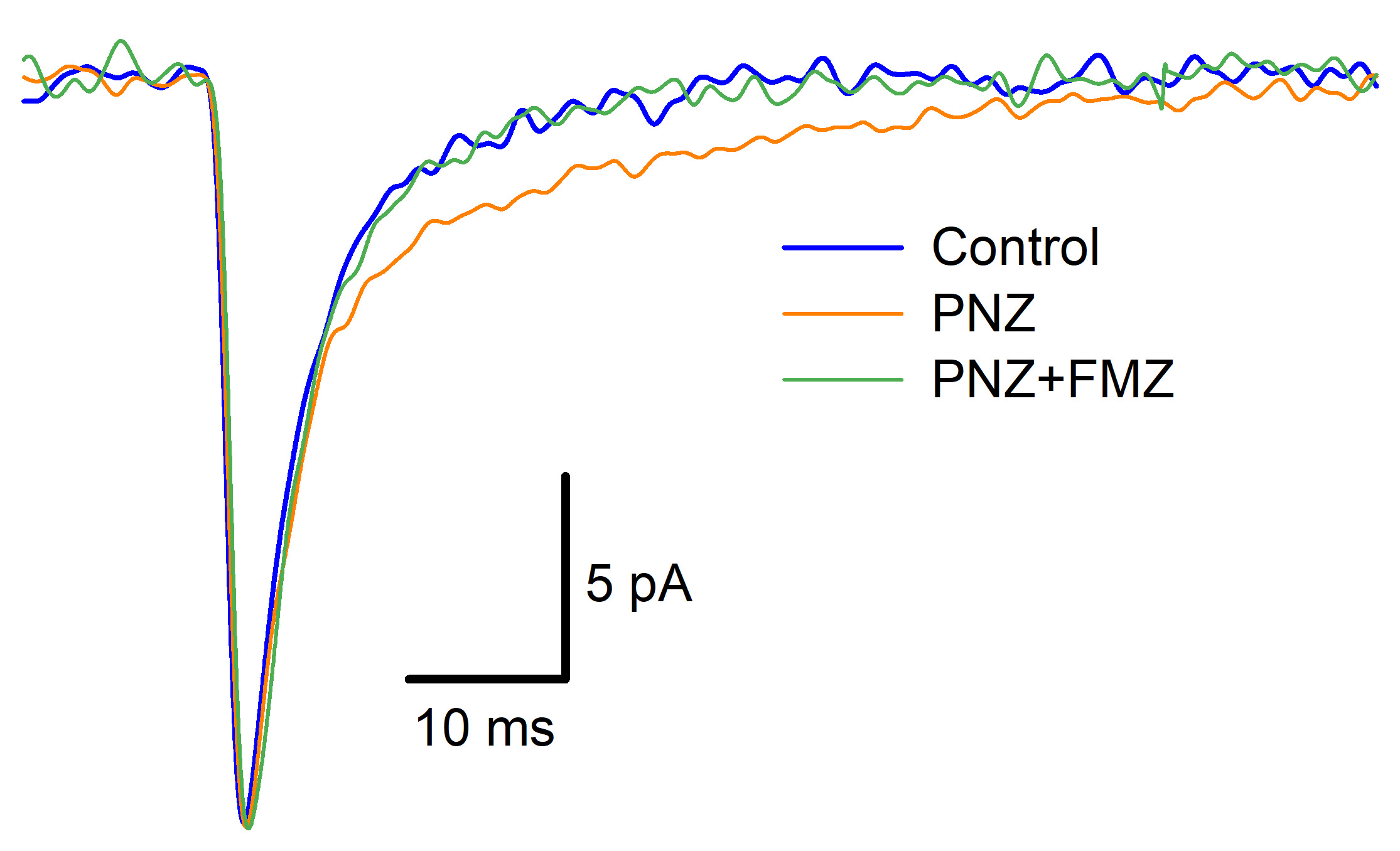
Species: Prussian carpAssay Type: In VitroVerified Customer | Posted 04/14/2020Soluble in DMSO, but requires sonication.Flumazenil (FMZ) was used to block action of other benzodiazepine ligand of GABA-A receptor (Phenazepam, PNZ) on decay kinetics of spontaneous inhibitory currents recorded from brain neurons of Prussian carp (Carassius gibelio). FMZ demonstrated competitive antagonism to PNZ in this research, both in vivo (behavioral tests) and in vitro (whole-cell voltage-clamp recordings, see example illustration).
-
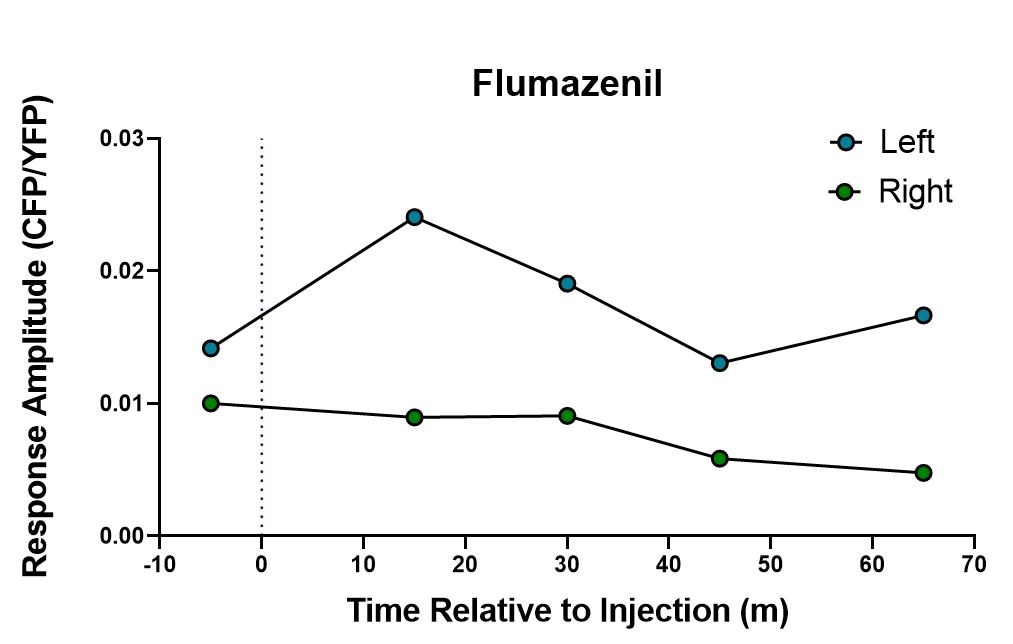
Species: MouseAssay Type: In VivoVerified Customer | Posted 05/23/2019injected 15mg/kg i.p at a concentration of.8mg/ml in 5% DMSO in salineused with fiber photometry to validate a chloride sensor in vivo. response to footshock was measured at baseline and then at 15 min intervals post injection
There are no reviews that match your criteria.