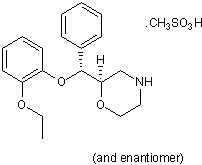
Reboxetine mesylate
Tocris Bioscience | Catalog # 1982

Key Product Details
Description
Product Description
Reboxetine mesylate is a potent and selective inhibitor of noradrenalin uptake (Ki values are 1.1, 129 and > 10000 nM for rat NET, SERT and DAT respectively). Displays > 1000-fold selectivity over α-adrenoceptors, 5-HT, dopamine and muscarinic ACh receptors. Orally active antidepressant.
Product Specifications for Reboxetine mesylate
Molecular Weight
Formula
Storage
Purity
Chemical Name
CAS Number
PubChem ID
InChI Key
SMILES
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Solubility
| Solvent | Max Conc. mg/mL | Max Conc. mM | |
|---|---|---|---|
| Solubility | |||
| water | 20.48 | 50 | |
| DMSO | 20.48 | 50 |
Preparing Stock Solutions for Reboxetine mesylate
The following data is based on the product molecular weight 409.50.
Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which all affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 0.5 mM | 4.88 mL | 24.42 mL | 48.84 mL |
| 2.5 mM | 0.98 mL | 4.88 mL | 9.77 mL |
| 5 mM | 0.49 mL | 2.44 mL | 4.88 mL |
| 25 mM | 0.10 mL | 0.49 mL | 0.98 mL |
Calculators
Background References
References are publications that support the biological activity of the product. See our Citations tab to view 51 publications citing the usage of this product.
- Owen and Whitton Reboxetine modulates NE efflux in the frontal cortex of the freely moving rat: the involvement of α2 and 5-HT1A receptors. Neurosci.Lett. 2003 PMID: 12932821
- Wong Reboxetine: a pharmacologically potent, selective, and specific NE reuptake inhibitor. Biol.Psychiatry. 2000 PMID: 10812041
- Millan S33005, a novel ligand at both serotonin and NE transporters: I Receptor binding, electrophysiological, and neurochemical profile in comparison with venlafaxine, reboxetine, citalopram, and clomipramine. J.Pharmacol.Exp.Ther. 2001 PMID: 11454918
- Sacchetti Studies on the acute and chronic effects of reboxetine on extracellular noradrenaline and other monoamines in the rat brain. Br.J.Pharmacol. 1999 PMID: 10578149
Product Documents for Reboxetine mesylate
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for Reboxetine mesylate
For research use only
Citations for Reboxetine mesylate
Customer Reviews for Reboxetine mesylate (1)
Have you used Reboxetine mesylate?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
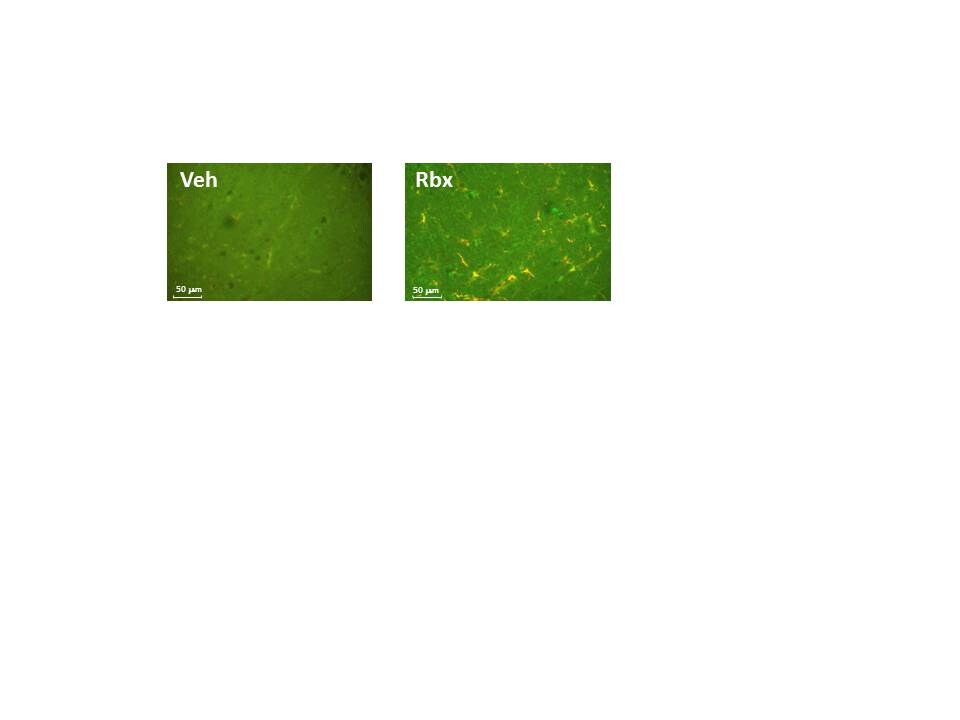
Species: MouseAssay Type: In VivoVerified Customer | Posted 01/10/2018In the figure, MCP-1 is labeled in red and GFAP in green.Reboxetine mesylate was dissolved in saline serum and 20 mg/kg (100 μL) were administered intraperitoneally to mice. After 24h, brain samples were taken and MCP-1 synthesis was analyzed by immunohistochemistry. This way, we confirmed that reboxetine treatment induces MCP-1 synthesis in cortical astrocytes.
There are no reviews that match your criteria.