DPPIV/CD26 plays an important role in many biological and pathological processes. It functions as T cell-activating molecule (THAM). It serves as a cofactor for entry of HIV in CD4+ cells. It binds adenosine deaminase, the deficiency of which causes severe combined immunodeficiency disease in humans. It cleaves chemokines such as stromal-cell-derived factor 1 alpha and macrophage-derived chemokine. It degrades peptide hormones such as glucagon. It truncates procalcitonin, a marker for systemic bacterial infections with elevated levels detected in patients with thermal injury, sepsis and severe infection, and in children with bacterial meningitis.

Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human, Mouse, Rat, Bat, Camelid, Ferret, Primate
Applications
Validated:
Immunohistochemistry, Western Blot, Immunocytochemistry, Simple Western
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Immunohistochemistry-Frozen, Western Blot, Neutralization, Flow Cytometry, Immunocytochemistry, Immunoprecipitation, Chromatin Immunoprecipitation (ChIP), Blocking
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant human DPPIV/CD26 (Catalog # 1180-SE)
Asp34-Pro766
Accession # Q53TN1
Asp34-Pro766
Accession # Q53TN1
Specificity
Detects human DPPIV/CD26 in direct ELISAs. In direct ELISAs, less than 5% cross-reactivity with recombinant mouse DPPIV is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human DPPIV/CD26 Antibody
Detection of Human DPPIV/CD26 by Western Blot.
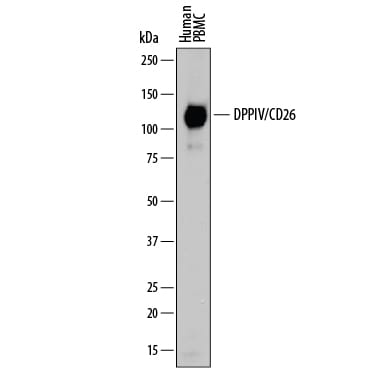
Western blot shows lysate of human peripheral blood mononuclear cells (PBMC). PVDF membrane was probed with 0.2 µg/mL of Goat Anti-Human DPPIV/CD26 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1180) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (HAF109). A specific band was detected for DPPIV/CD26 at approximately 110 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Detection of Human DPPIV/CD26 by Western Blot.
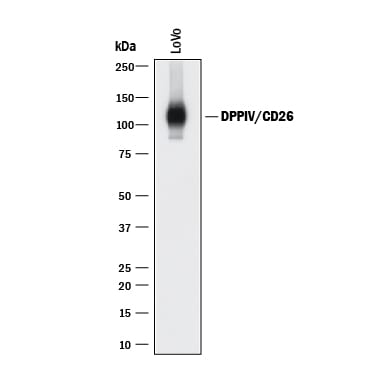
Western blot shows lysate of LoVo human colorectal adenocarcinoma cell line. PVDF membrane was probed with 0.2 µg/mL of Goat Anti-Human DPPIV/CD26 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1180) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (HAF017). A specific band was detected for DPPIV/CD26 at approximately 110 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.DPPIV/CD26 in Human PBMCs.
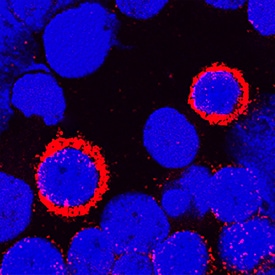
DPPIV/CD26 was detected in immersion fixed human peripheral blood mononuclear cells (PBMCs) using Goat Anti-Human DPPIV/CD26 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1180) at 1.7 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; NL001) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm and plasma membranes. View our protocol for Fluorescent ICC Staining of Non-adherent Cells.DPPIV/CD26 in Human Psoriatic Skin.
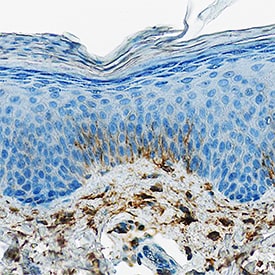
DPPIV/CD26 was detected in immersion fixed paraffin-embedded sections of human psoriatic skin using Goat Anti-Human DPPIV/CD26 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1180) at 1 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; CTS008) and counterstained with hematoxylin (blue). Specific staining was localized to keratinocytes. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Detection of Human DPPIV/CD26 by Simple WesternTM.
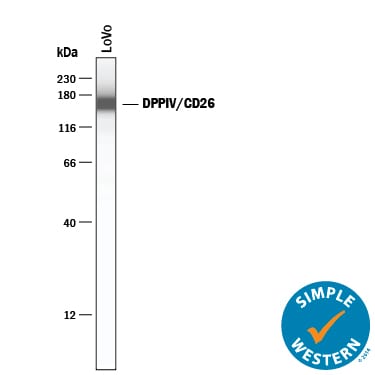
Simple Western lane view shows lysates of LoVo human colorectal adenocarcinoma cell line, loaded at 0.2 mg/mL. A specific band was detected for DPPIV/CD26 at approximately 168 kDa (as indicated) using 2 µg/mL of Goat Anti-Human DPPIV/CD26 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1180) followed by 1:50 dilution of HRP-conjugated Anti-Goat IgG Secondary Antibody (HAF109). This experiment was conducted under reducing conditions and using the 12-230 kDa separation system.Detection of Human DPPIV/CD26 by Western Blot
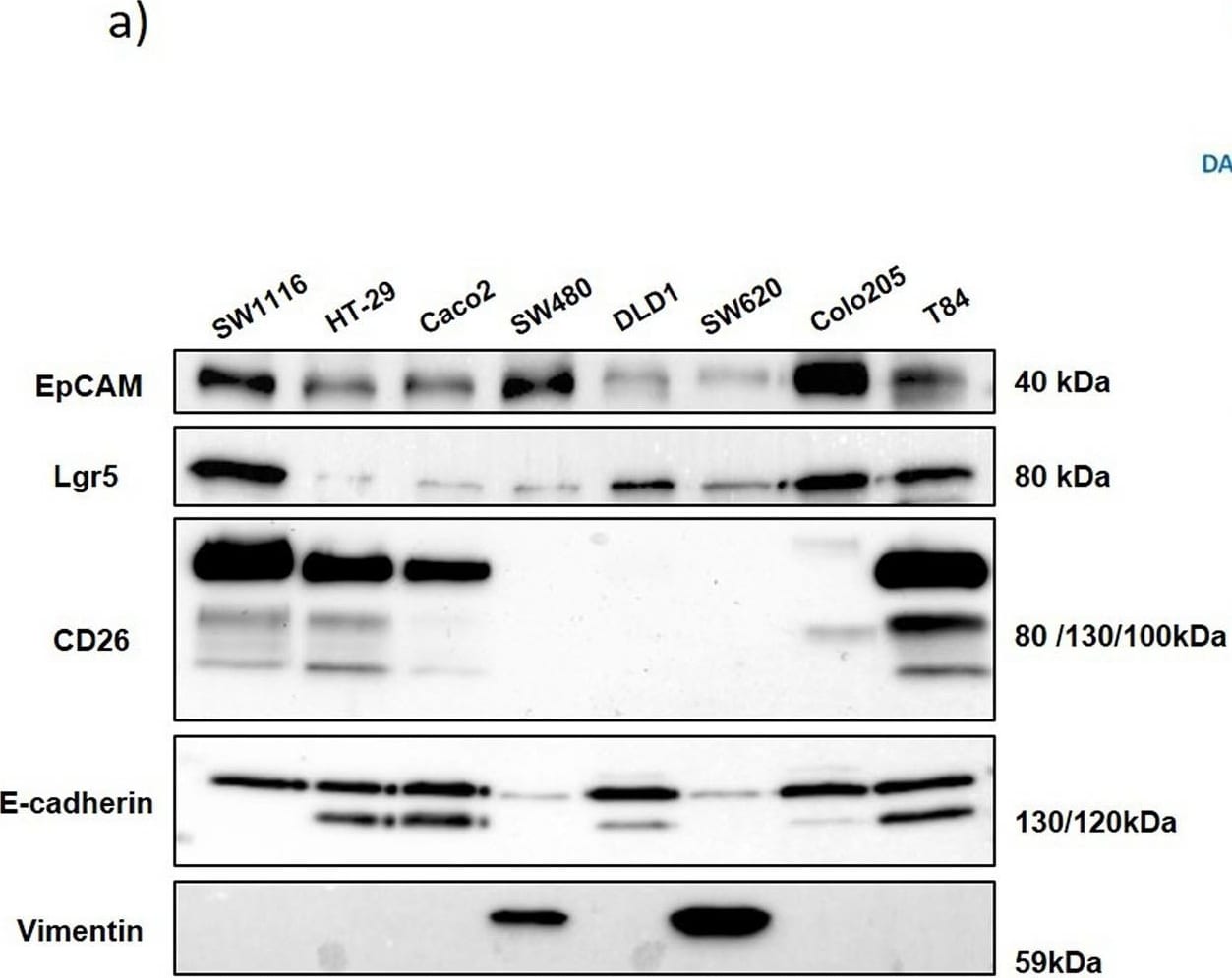
Expression of different markers in the eight human colon cancer cell lines analyzed. (A) Western blot analysis of EpCAM, LGR5, CD26, E-cadherin and vimentin expression in total cell extracts from the eight cell lines (20 μg of protein in each line). Data shown are representative of three experiments. (B) E-cadherin and EpCAM expression analysis by immunofluorescence in HT-29 and Caco-2 cells. (C) CD44 and CD26 expression analysis by immunofluorescence in HT-29 and Caco-2 cells. (D) LGR5 expression analysis by immunofluorescence in DLD-1 and Caco-2 cells. Nuclei were stained with DAPI. Scale bars: 50 μm. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/31285270), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human DPPIV/CD26 by Immunocytochemistry/ Immunofluorescence
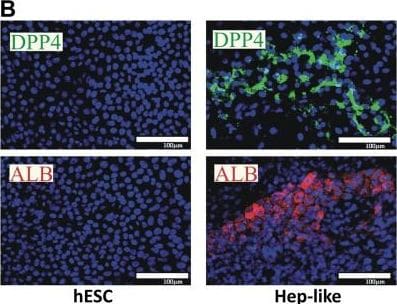
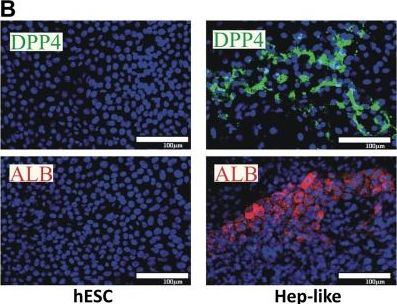
Comparison of histone modifier sets between two hepatic cell cultures.(A) Differentiation scheme of hESC towards Hepatocyte-like cells (Hep-like). (B) Hep-like cells were generated from hESC and stained with antibodies specific for DPP4 and albumin. Nuclei were stained with the DNA dye H-33342 (blue). Scale bars: 100 µm. (C) RT-qPCR data from three independent experiments for hepatic (ALB, CYP3A, CYP7A1, DPP4, HNF4, MET) and neuronal (TH, DCX, TUBB3) markers. Relative gene expression was calculated using hESC as a calibrator and a set of three reference genes (HPRT, RPL13A, GAPDH). (D) Transcript levels of epigenetic modifiers were measured by RT-qPCR in human liver (huHep), Hep-like islets (Hep-like) and embryonic stem cells (hESC). Data for huHep and Hep-like are indicated as relative change compared to hESC (as reference cell). For comparative display, a scatter plot was constructed so that differentially expressed genes that show pos. association (between Hep-like and huHep) are found in red fields, and those that differed in the sense of regulation fall into blue fields. Values of >10 were set to 10. For quadrant count ratio analysis (QCR) only expression values >2 or <−2 were included. (E) The data measured in D were plotted as heat map, sorted according to rel. huHep expression levels. Transcripts that were >2-fold higher expressed in tissue than in hESC are marked in red, >2-fold lower expression is marked in blue. The color scale ranges from a fold regulation of −20 (dark blue) to +20 (dark red). Measures of variance and p-values are indicated in the supplemental material, genes not regulated significantly (vs. hESC) are displayed as “n.s.”. Image collected and cropped by CiteAb from the following open publication (https://dx.plos.org/10.1371/journal.pone.0102035), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human DPPIV/CD26 by Immunocytochemistry/ Immunofluorescence
Comparison of histone modifier sets between two hepatic cell cultures.(A) Differentiation scheme of hESC towards Hepatocyte-like cells (Hep-like). (B) Hep-like cells were generated from hESC and stained with antibodies specific for DPP4 and albumin. Nuclei were stained with the DNA dye H-33342 (blue). Scale bars: 100 µm. (C) RT-qPCR data from three independent experiments for hepatic (ALB, CYP3A, CYP7A1, DPP4, HNF4, MET) and neuronal (TH, DCX, TUBB3) markers. Relative gene expression was calculated using hESC as a calibrator and a set of three reference genes (HPRT, RPL13A, GAPDH). (D) Transcript levels of epigenetic modifiers were measured by RT-qPCR in human liver (huHep), Hep-like islets (Hep-like) and embryonic stem cells (hESC). Data for huHep and Hep-like are indicated as relative change compared to hESC (as reference cell). For comparative display, a scatter plot was constructed so that differentially expressed genes that show pos. association (between Hep-like and huHep) are found in red fields, and those that differed in the sense of regulation fall into blue fields. Values of >10 were set to 10. For quadrant count ratio analysis (QCR) only expression values >2 or <−2 were included. (E) The data measured in D were plotted as heat map, sorted according to rel. huHep expression levels. Transcripts that were >2-fold higher expressed in tissue than in hESC are marked in red, >2-fold lower expression is marked in blue. The color scale ranges from a fold regulation of −20 (dark blue) to +20 (dark red). Measures of variance and p-values are indicated in the supplemental material, genes not regulated significantly (vs. hESC) are displayed as “n.s.”. Image collected and cropped by CiteAb from the following open publication (https://dx.plos.org/10.1371/journal.pone.0102035), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of DPPIV/CD26 by Western Blot
Nuclear Translocation of Antitumor CD26 mAbs in Cancer Cells. (D) Jurkat/mock or Jurkat/CD26 cells incubated with biotin-labeled control IgG1 or 1F7 for 1 hour. Nuclear (Nuc)&membrane (Mem) extracts of these cells pulled-down with Neutravidin,&then subjected to immunoblot analysis using streptavidin or antibodies to CD26 or Na+/K+ ATPase (membrane marker). HC, heavy chain; LC, light chain. Image collected & cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/23638030), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human DPPIV/CD26 Antibody
Application
Recommended Usage
Immunocytochemistry
1-15 µg/mL
Sample: Immersion fixed human peripheral blood mononuclear cells (PBMCs)
Sample: Immersion fixed human peripheral blood mononuclear cells (PBMCs)
Immunohistochemistry
1-15 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human prostate and psoriatic skin
Sample: Immersion fixed paraffin-embedded sections of human prostate and psoriatic skin
Simple Western
2 µg/mL
Sample: LoVo human colorectal adenocarcinoma cell line
Sample: LoVo human colorectal adenocarcinoma cell line
Western Blot
0.2 µg/mL
Sample: Human peripheral blood mononuclear cells (PBMC) and LoVo human colorectal adenocarcinoma cell line
Sample: Human peripheral blood mononuclear cells (PBMC) and LoVo human colorectal adenocarcinoma cell line
Reviewed Applications
Read 6 reviews rated 4.7 using AF1180 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. See Certificate of Analysis for details.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: DPPIV/CD26
Long Name
Dipeptidyl-peptidase IV
Alternate Names
CD26, DPP4
Gene Symbol
DPP4
UniProt
Additional DPPIV/CD26 Products
Product Documents for Human DPPIV/CD26 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human DPPIV/CD26 Antibody
For research use only
Citations for Human DPPIV/CD26 Antibody
Customer Reviews for Human DPPIV/CD26 Antibody (6)
4.7 out of 5
6 Customer Ratings
Have you used Human DPPIV/CD26 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
5 的
6 reviews
Showing All
Filter By:
-
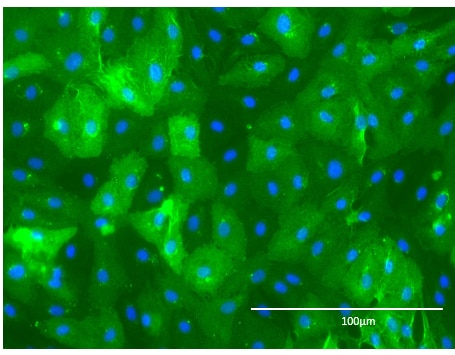
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: HMVEC human microvascular endothelial cellsSpecies: HumanVerified Customer | Posted 01/30/2023
-
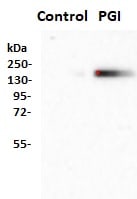
Application: Western BlotSample Tested: HepG2 human hepatocellular carcinoma cell lineSpecies: HumanVerified Customer | Posted 10/09/2020Serum-starved HepG2 cells were treated with 100 ng/ml phosphoglucose isomerase (PGI) in minimal essential medium for 2 days, following which cells were harvested and analyzed by Western blotting using the CD26 antibody (1000 fold dilution). The study shows that PGI induces the expression of CD26.
-
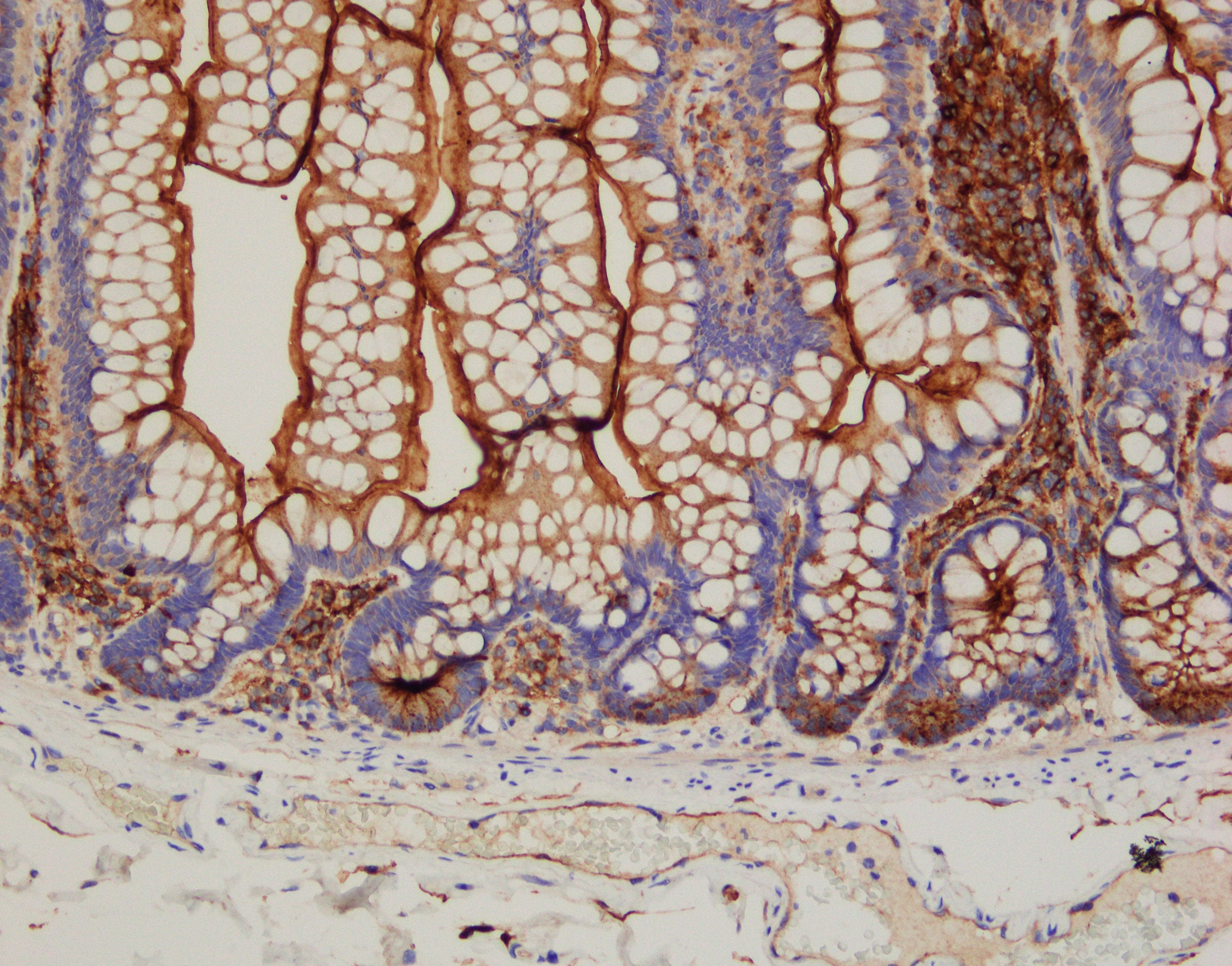
Application: ImmunohistochemistrySample Tested: Small intestineSpecies: HumanVerified Customer | Posted 01/21/2020IHC-paraffin working dilution: 1ug/mL with Bond Refine polymer Detection rabbit anti-goat IgG as a Linker antibody epitope retrieval: basic (BOND ER2)
-
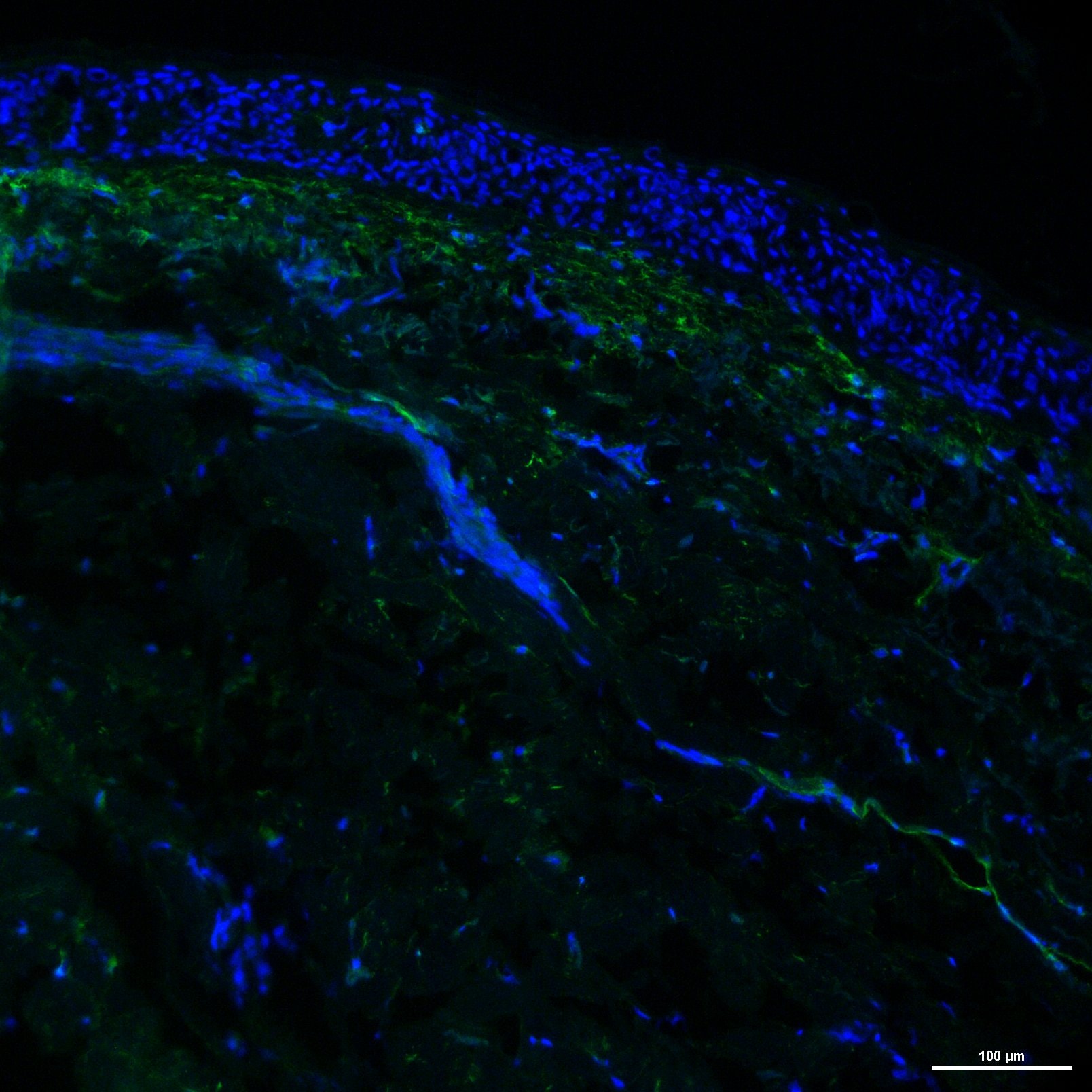
Application: Immunohistochemistry-FrozenSample Tested: Frozen human skin sectionsSpecies: HumanVerified Customer | Posted 02/22/2019
-
Application: ImmunoprecipitationSample Tested: Heparin PlasmaSpecies: MouseVerified Customer | Posted 08/11/2016
-
Application: Immunohistochemistry-ParaffinSample Tested: See PMID 23638030Species: HumanVerified Customer | Posted 01/05/2015
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...