Pyridostatin pentahydrochloride
Tocris Bioscience | Catalog # 4763

Key Product Details
Description
Product Description
Binds and stabilizes G-quadruplexes, inducing DNA damage and cell cycle arrest (Kd = 490 nM); targets the proto-oncogene Src, reducing Src protein abundance and Src-dependent motility in human breast cancer cells. Also targets telomeric G-quadruplexes, inducing telomerase dysfunction. Activates the DNA-dependent protein kinase catalytic sunbunit (DNA-PKcs).
Product Specifications for Pyridostatin pentahydrochloride
Molecular Weight
Formula
Storage
Purity
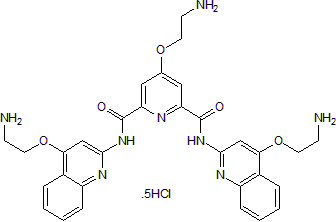
Chemical Name
CAS Number
PubChem ID
InChI Key
SMILES
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Solubility
| Solvent | Max Conc. mg/mL | Max Conc. mM | |
|---|---|---|---|
| Solubility | |||
| water | 15.58 | 20 |
Preparing Stock Solutions for Pyridostatin pentahydrochloride
The following data is based on the product molecular weight 778.94.
Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which all affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 0.2 mM | 6.42 mL | 32.09 mL | 64.19 mL |
| 1 mM | 1.28 mL | 6.42 mL | 12.84 mL |
| 2 mM | 0.64 mL | 3.21 mL | 6.42 mL |
| 10 mM | 0.13 mL | 0.64 mL | 1.28 mL |
Calculators
Background References
References are publications that support the biological activity of the product. See our Citations tab to view 6 publications citing the usage of this product.
- Müller Small-molecule-mediated G-quadruplex isolation from human cells. Nat.Chem. 2010 PMID: 21107376
- Koirala A single-molecule platform for investigation of interactions between G-quadruplexes and small-molecule ligands. Nat.Chem. 2011 PMID: 21941250
- Rodriguez Small-molecule-induced DNA damage identifies alternative DNA structures in human genes. Nat.Chem.Biol. 20112 PMID: 22306580
Product Documents for Pyridostatin pentahydrochloride
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for Pyridostatin pentahydrochloride
For research use only
Citations for Pyridostatin pentahydrochloride
Customer Reviews for Pyridostatin pentahydrochloride
There are currently no reviews for this product. Be the first to review Pyridostatin pentahydrochloride and earn rewards!
Have you used Pyridostatin pentahydrochloride?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
