Having trouble trying to find the right product? Chat with our scientists below - we're here to help!
251 results for "FGFR5/FGFRL1" in 产品
FGFR5/FGFRL1 Products
FGF activity is mediated by a family of type I transmembrane tyrosine kinases, which undergo dimerization and autophosphorylation after ligand binding. Five distinct genes encode closely related FGF receptors, FGFR1 through 5. FGFRs contain three Ig-like domains and a stretch of acidic residues between the first and second Ig-like domains. FGFR1, 2, 3, and -4 have a cytoplasmic split tyrosine-kinase domain, but FGFR5 does not. Multiple forms of FGFR1, 2, and 3 are generated by alternative splicing.
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Mouse |
| Source: | NS0 |
| Accession #: | NP_473412 |
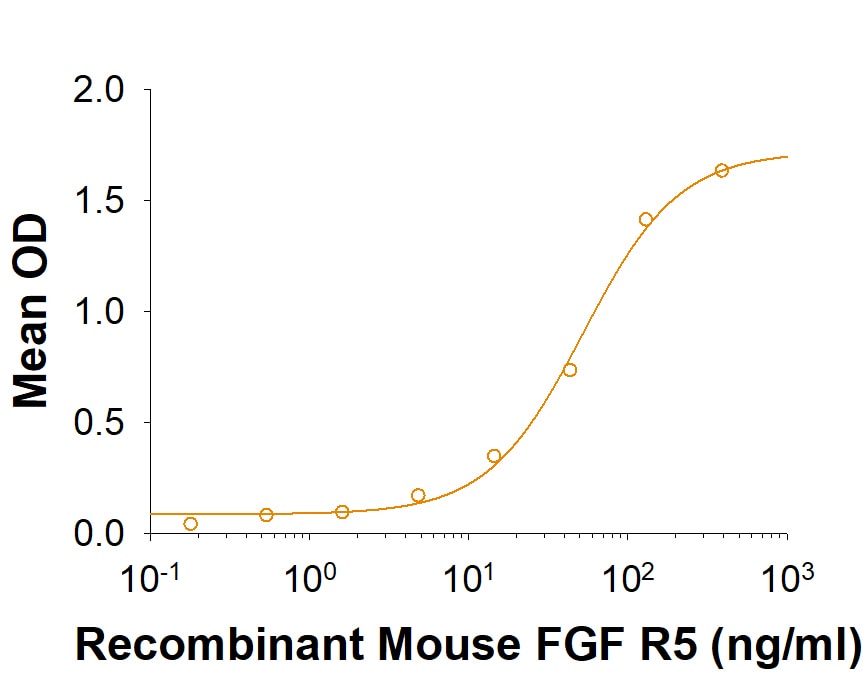
| Applications: | Bioactivity |
Loading...
| Reactivity: | Human |
| Source: | HEK293 |
| Accession #: | Q8N441 |
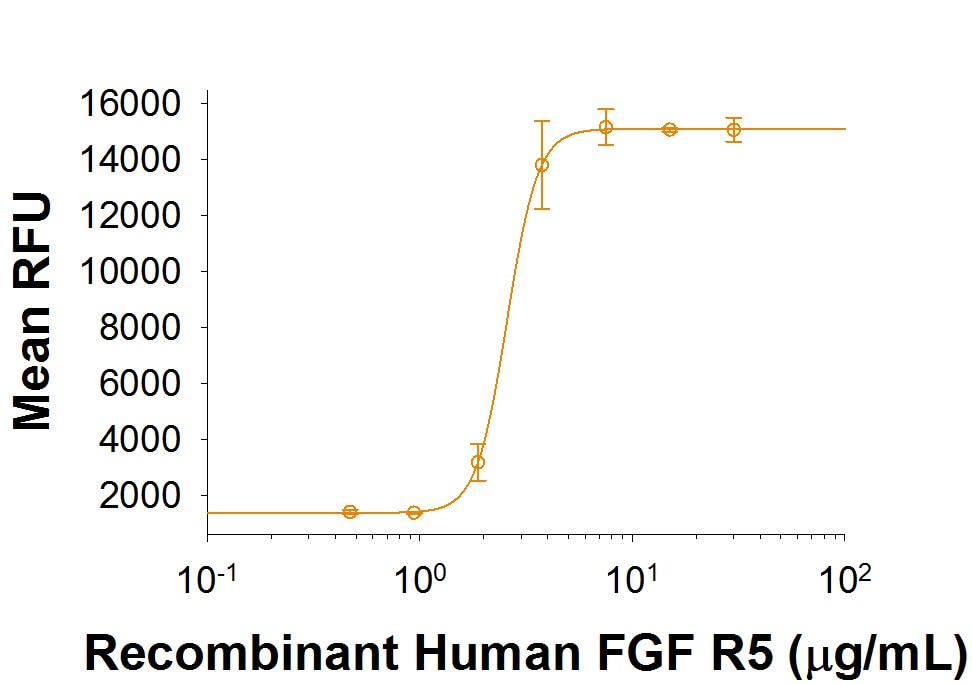
| Applications: | Bioactivity |
Loading...
| Reactivity: | Mouse |
| Details: | Rat IgG2B Monoclonal Clone #223003 |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Human |
Loading...
| Reactivity: | Mouse |
Loading...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
Recombinant Monoclonal Antibody
| Reactivity: | Mouse |
| Details: | Rabbit IgG Monoclonal Clone #019 |
| Applications: | WB, IP |
Loading...
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | WB |
Loading...


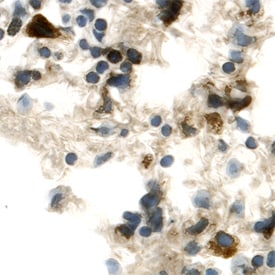

![Western Blot: FGFR5/FGFRL1 Antibody [NBP1-31461] Western Blot: FGFR5/FGFRL1 Antibody [NBP1-31461]](https://resources.rndsystems.com/images/products/FGFR5-FGFRL1-Antibody-Western-Blot-NBP1-31461-img0013.jpg)

![Western Blot: FGFR5/FGFRL1 Antibody (019) [NBP2-90467] Western Blot: FGFR5/FGFRL1 Antibody (019) [NBP2-90467]](https://resources.rndsystems.com/images/products/FGFR5-FGFRL1-Antibody-019-Western-Blot-NBP2-90467-img0003.jpg)
![Immunocytochemistry/ Immunofluorescence: FGFR5/FGFRL1 Antibody [NBP2-56879] Immunocytochemistry/ Immunofluorescence: FGFR5/FGFRL1 Antibody [NBP2-56879]](https://resources.rndsystems.com/images/products/FGF-R5-FGFRL1-Antibody-Immunocytochemistry-Immunofluorescence-NBP2-56879-img0001.jpg)