Having trouble trying to find the right product? Chat with our scientists below - we're here to help!
14 results for "PAR4" in 产品
PAR4 Products
PAR4, also known as F2RL3 and Coagulation Factor II (Thrombin) Receptor-like 3, is a member of the Protease-activated Receptor (PAR) family of seven transmembrane G protein-coupled receptors that are activated by proteolysis. It is 385 amino acids (aa) in length with a predicted molecular weight of 41.1 kDa. Human PAR4 shares 78% aa sequence identity with the mouse ortholog. PAR4 is widely expressed with highest expression levels being found in the lungs, pancreas, thyroid, testis, and small ...
Selective PAR4 agonist
| Alternate Names: | AYPGKF-NH2 |
| Purity: | ≥95% |
Loading...
| Reactivity: | Human |
Loading...
| Reactivity: | Mouse |
Loading...
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | WB, ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG1 kappa Monoclonal Clone #5F4 |
| Applications: | WB, ELISA, Flow |
Loading...
| Reactivity: | Human, Mouse, Rat |
| Details: | Rabbit IgG Polyclonal |
| Applications: | WB, ELISA |
Loading...
| Reactivity: | Human |
| Source: | Wheat germ |
| Applications: | WB, ELISA, AP, MA |
Loading...
| Reactivity: | Human |
| Sensitivity: | 0.19 ng/mL |
| Assay Range: | 0.31 - 20 ng/mL |
| Applications: | ELISA |
Loading...
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | ICC/IF |
Loading...
| Reactivity: | Human |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC |
Loading...
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC |
Loading...
| Reactivity: | Human |
| Source: | E. coli |
| Applications: | AC |
Loading...
| Reactivity: | Human |
| Source: | E. coli |
| Applications: | AC |
Loading...


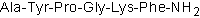



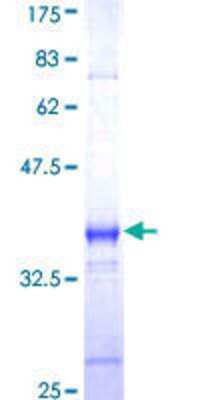
![Western Blot: PAR4 Antibody [NBP3-21255] - Western Blot: PAR4 Antibody [NBP3-21255] -](https://resources.rndsystems.com/images/products/nbp3-21255_rabbit-par4-pab-45202315493266.jpg)
![ELISA: PAR4 Antibody (5F4) [H00009002-M01] ELISA: PAR4 Antibody (5F4) [H00009002-M01]](https://resources.rndsystems.com/images/products/PAR4-Antibody-5F4-ELISA-H00009002-M01-img0003.jpg)
![Western Blot: PAR4 Antibody [NBP3-38550] - PAR4 Antibody](https://resources.rndsystems.com/images/products/nbp3-38550_rabbit-par4-pab-1012202411353024.jpg)
![ELISA: Human PAR4 ELISA Kit (Colorimetric) [NBP3-06971] - Human PAR4 ELISA Kit (Colorimetric)](https://resources.rndsystems.com/images/products/nbp3-06971_human-par4-elisa-kit-colorimetric-132202416251795.jpg)
![Immunocytochemistry/ Immunofluorescence: PAR4 Antibody [NBP3-17410] Immunocytochemistry/ Immunofluorescence: PAR4 Antibody [NBP3-17410]](https://resources.rndsystems.com/images/products/PAR4-Antibody-Immunocytochemistry-Immunofluorescence-NBP3-17410-img0001.jpg)
![Western Blot: PAR4 Overexpression Lysate [NBL1-10411] Western Blot: PAR4 Overexpression Lysate [NBL1-10411]](https://resources.rndsystems.com/images/products/Proteinase-activated-receptor-4-PAR4-Overexpression-Lysate-Adult-Normal-Western-Blot-NBL1-10411-img0002.jpg)
![Immunohistochemistry-Paraffin: PAR4 Antibody - BSA Free [NLS1308] Immunohistochemistry-Paraffin: PAR4 Antibody - BSA Free [NLS1308]](https://resources.rndsystems.com/images/products/PAR4-Antibody-Immunohistochemistry-Paraffin-NLS1308-img0001.jpg)
![Immunohistochemistry-Paraffin: PAR4 Antibody - BSA Free [NLS1311] Immunohistochemistry-Paraffin: PAR4 Antibody - BSA Free [NLS1311]](https://resources.rndsystems.com/images/products/PAR4-Antibody-Immunohistochemistry-Paraffin-NLS1311-img0001.jpg)