ABT 199
Tocris Bioscience | Catalog # 6960

Key Product Details
Description
Alternative Names
Product Description
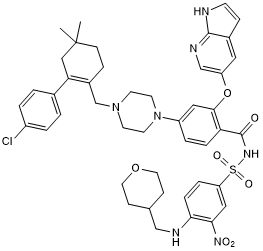
ABT 199 is a selective, high affinity Bcl-2 inhibitor (Ki < 0.010 nM). Exhibits >4800-fold selectivity for Bcl-2 over Bcl-xL and Bcl-w, and displays no measurable activity at Mcl-1 (Ki > 444 nM). Potently induces apoptosis in FL5.12-BCL-2 cells (EC50 = 261 nM) and reduces tumor burden in chronic lymphocytic leukemia (CLL) primary samples (EC50 = 3 nM). Shows reduced toxicity to platelets compared to similar compounds. Enhances efficacy of clinically relevant chemotherapy and immunotherapy drugs. Orally bioavailable. Exhibits binding to SARS-CoV-2 3C-like protease (3CLpro) active site in a virtual screen. ABT 199 reverses oxidative phosphorylation in acute myeloid leukemia cells (AML), in vitro and in vivo.
Product Specifications for ABT 199
Molecular Weight
Formula
Storage
Purity
Chemical Name
CAS Number
PubChem ID
InChI Key
SMILES
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Solubility
| Solvent | Max Conc. mg/mL | Max Conc. mM | |
|---|---|---|---|
| Solubility | |||
| DMSO | 86.84 | 100 |
Preparing Stock Solutions for ABT 199
The following data is based on the product molecular weight 868.45.
Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which all affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 1 mM | 1.15 mL | 5.76 mL | 11.51 mL |
| 5 mM | 0.23 mL | 1.15 mL | 2.30 mL |
| 10 mM | 0.12 mL | 0.58 mL | 1.15 mL |
| 50 mM | 0.02 mL | 0.12 mL | 0.23 mL |
Calculators
Background References
References are publications that support the biological activity of the product. See our Citations tab to view 14 publications citing the usage of this product.
- Bosc Mitochondrial inhibitors circumvent adaptive resistance to venetoclax and cytarabine combination therapy in acute myeloid leukemia. Nat.cancer 2021 PMID: 35122057
- Soderquist Systematic mapping of BCL-2 gene dependencies in cancer reveals molecular determinants of BH3 mimetic sensitivity. Nat.Commun. 2018 PMID: 30158527
- Liu Balancing apoptosis and autophagy for Parkinson's disease therapy: targeting BCL-2. ACS Chem.Neurosci. 2019 PMID: 30400738
- Souers ABT-199, a potent and selective BCL-2 inhibitor, achieves antitumor activity while sparing platelets. Nat.Med. 2013 PMID: 23291630
Product Documents for ABT 199
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for ABT 199
For research use only
Related Research Areas
Citations for ABT 199
Customer Reviews for ABT 199
There are currently no reviews for this product. Be the first to review ABT 199 and earn rewards!
Have you used ABT 199?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
