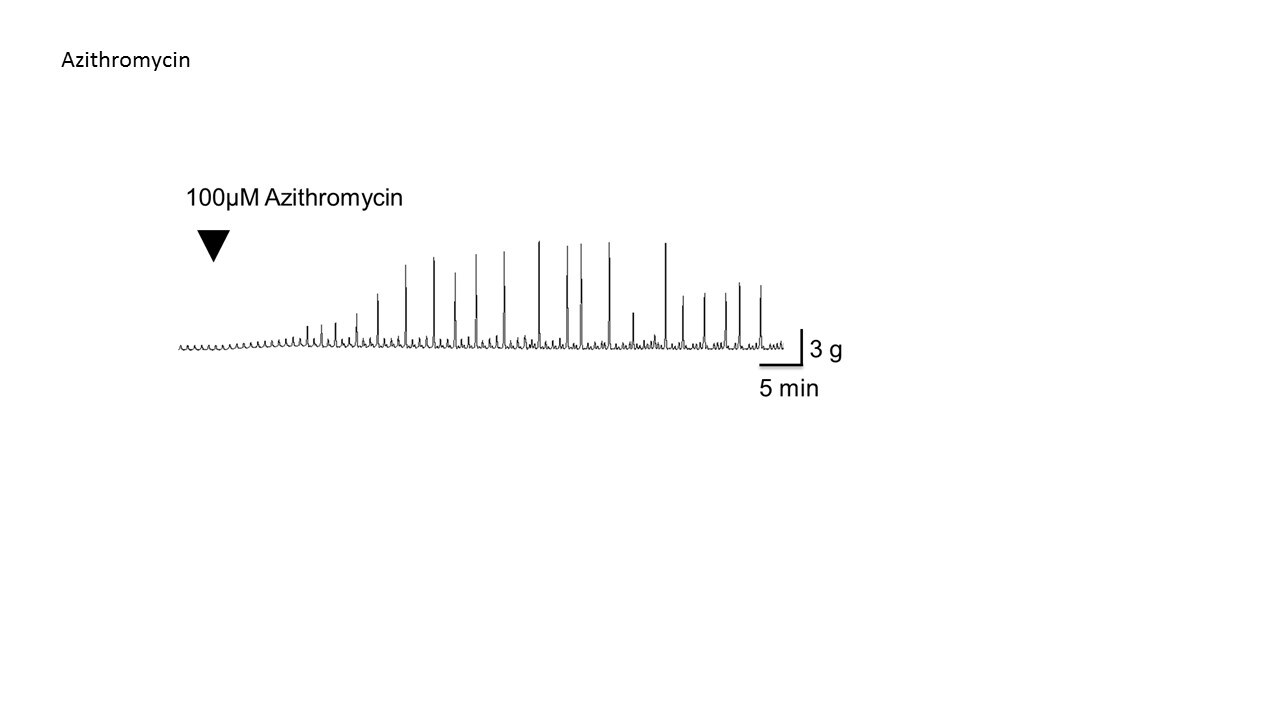
Azithromycin
Tocris Bioscience | Catalog # 3771

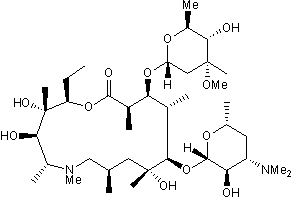
Key Product Details
Description
Alternative Names
Product Description
Azithromycin is a macrolide antibiotic. Inhibits 50S ribosomal subunit formation and elongation at transpeptidation step in gram-positive and gram-negative organisms. Orally active with improved pharmacokinetics over erythromycin in mouse models. Inhibits autophagy. Predicted to disrupt binding of SARS-CoV-2 spike protein to ACE2.Licensing Information
Sold for research purposes under agreement from Pfizer Inc.
Product Specifications for Azithromycin
Molecular Weight
Formula
Storage
Purity
Chemical Name
CAS Number
PubChem ID
InChI Key
SMILES
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Solubility
| Solvent | Max Conc. mg/mL | Max Conc. mM | |
|---|---|---|---|
| Solubility | |||
| DMSO | 74.9 | 100 | |
| Ethanol | 74.9 | 100 |
Preparing Stock Solutions for Azithromycin
The following data is based on the product molecular weight 748.98.
Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which all affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 1 mM | 1.34 mL | 6.68 mL | 13.35 mL |
| 5 mM | 0.27 mL | 1.34 mL | 2.67 mL |
| 10 mM | 0.13 mL | 0.67 mL | 1.34 mL |
| 50 mM | 0.03 mL | 0.13 mL | 0.27 mL |
Calculators
Background References
References are publications that support the biological activity of the product. See our Citations tab to view 16 publications citing the usage of this product.
- Sandeep and McGregor Energetics based modeling of hydroxychloroquine and azithromycin binding to the SARS-CoV-2 spike (S) protein - ACE2 complex . ChemRxiv - Paper not yet peer reviewed.
- Galluzzi Pharmacological modulation of autophagy: therapeutic potential and persisting obstacles. Nat.Rev.Drug.Discov. 2017 PMID: 28529316
- Girard Pharmacokinetic and in vivo studies with azithro. (CP-62,993), a new macrolide with extended half-life and excellent tissue distribution. Antimicrob.Agents Chemother. 1987 PMID: 2830841
- Champney and Burdine Macrolide antibiotics inhibit 50S ribosomal subunit assembly in Bacillus subtilis and Staphylococcus aureas. Antimicrob.Agents Chemother. 1995 PMID: 8540733
- Retsema Spectrum and mode of action of azithro. (CP-62,993), a new 15-membered-ring macrolide with improved potency against gram-negative organisms. Antimicrob.Agents Chemother. 1987 PMID: 2449865
Product Documents for Azithromycin
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for Azithromycin
For research use only
Citations for Azithromycin
Customer Reviews for Azithromycin (1)
Have you used Azithromycin?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Species: HumanAssay Type: In VitroVerified Customer | Posted 01/05/2018Use at 100-300uM for efficacy as motilin agonistHuman EFS GI smooth muscle assay
There are no reviews that match your criteria.