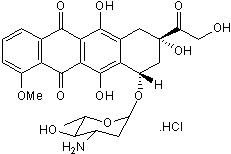
Epirubicin hydrochloride
Tocris Bioscience | Catalog # 3260

Key Product Details
Description
Alternative Names
Product Description
Epirubicin hydrochloride is an antibiotic antitumor agent. Inhibits the synthesis and function of DNA (IC50 = 62.7 μM in rat glioblastoma cell lines) and inhibits the relaxing property of topoisomerase II.
Product Specifications for Epirubicin hydrochloride
Molecular Weight
Formula
Storage
Purity
Chemical Name
CAS Number
PubChem ID
InChI Key
SMILES
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Solubility
| Solvent | Max Conc. mg/mL | Max Conc. mM | |
|---|---|---|---|
| Solubility | |||
| water | 29 | 50 | |
| DMSO | 58 | 100 |
Preparing Stock Solutions for Epirubicin hydrochloride
The following data is based on the product molecular weight 579.98.
Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which all affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 1 mM | 1.72 mL | 8.62 mL | 17.24 mL |
| 5 mM | 0.34 mL | 1.72 mL | 3.45 mL |
| 10 mM | 0.17 mL | 0.86 mL | 1.72 mL |
| 50 mM | 0.03 mL | 0.17 mL | 0.34 mL |
Calculators
Background References
References are publications that support the biological activity of the product.
- Spadari DNA polymerases and DNA topoisomerases as targets for the development of anticancer drugs. Anticancer Res. 1986 PMID: 3026237
- Schott and Robert Comparative activity of anthracycline 13-dihydrometabolites against rat glioblastoma cells in culture. Biochem.Pharmacol. 1989 PMID: 2597184
- Cersosimo Epirubicin: a review of pharmacology, clinical activity, and adverse effects of an adriamycin analogue. J.Clin.Oncol. 1986 PMID: 3005521
Product Documents for Epirubicin hydrochloride
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for Epirubicin hydrochloride
For research use only
Citations for Epirubicin hydrochloride
Customer Reviews for Epirubicin hydrochloride (2)
Have you used Epirubicin hydrochloride?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
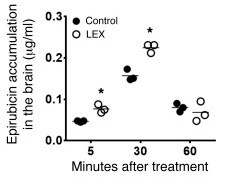
-
Species: MouseAssay Type: In VivoCell Line/Tissue: brainVerified Customer | Posted 05/09/202010 mg/kg of epirubicin
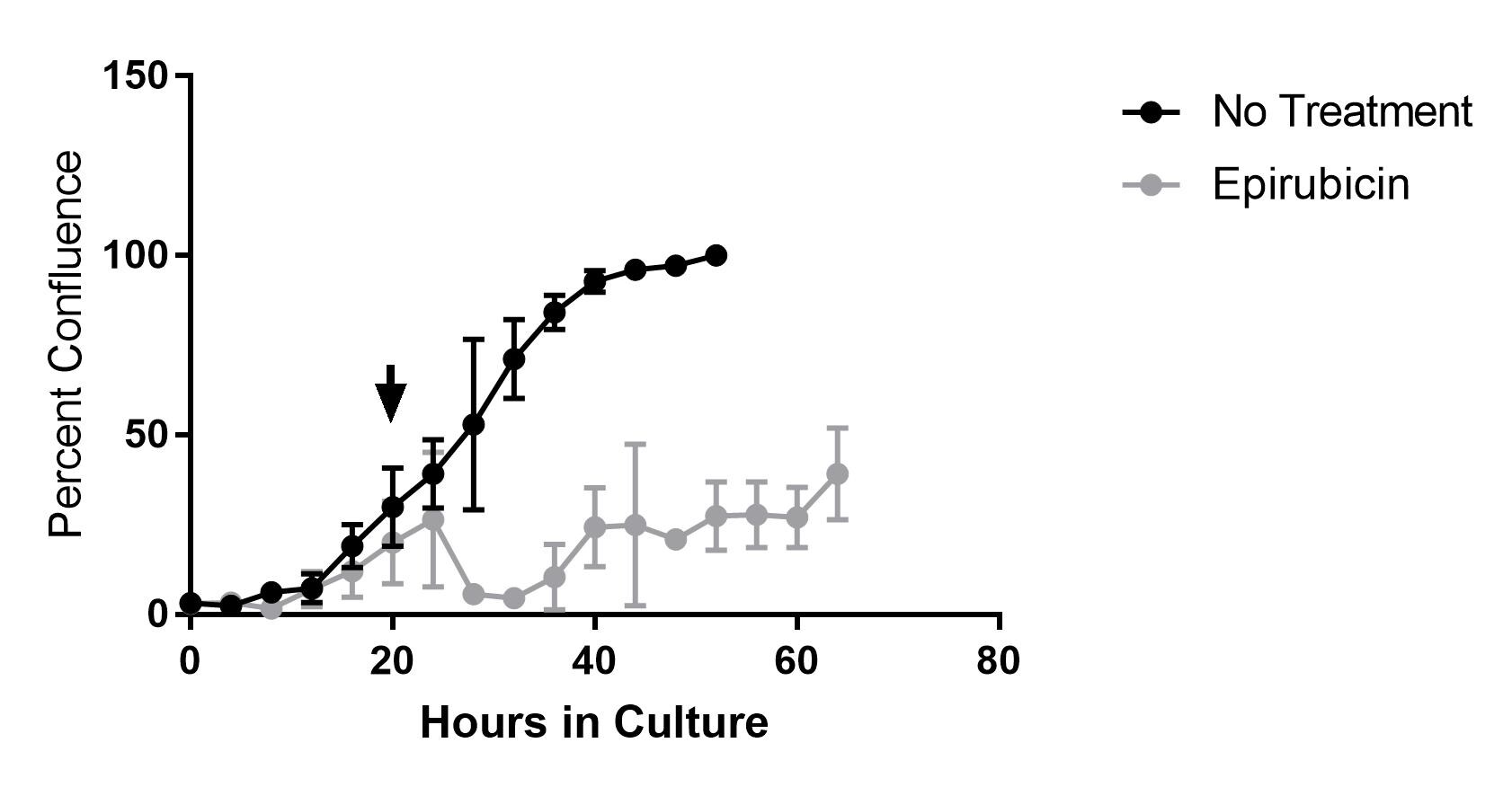
-
Species: HumanAssay Type: In VitroVerified Customer | Posted 02/14/2019Epirubicin was used at a 20 uM concentration for in vitro culture with SK-MEL-28 and cell viability was monitored over time with live imaging. Arrow indicates when compound was added to media.
There are no reviews that match your criteria.