H2L5186303
Tocris Bioscience | Catalog # 4878

Key Product Details
Description
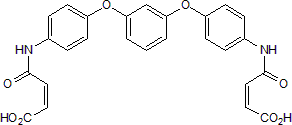
Product Description
H2L5186303 is a potent and selective lysophosphatidic acid 2 (LPA2) receptor antagonist (IC50 values are 8.9, 1230 and 27354 nM for LPA2, LPA3 and LPA1 receptors respectively, in a LPA-elicited calcium mobilization assay).
Product Specifications for H2L5186303
Molecular Weight
Formula
Storage
Purity
Chemical Name
CAS Number
PubChem ID
InChI Key
SMILES
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Solubility
| Solvent | Max Conc. mg/mL | Max Conc. mM | |
|---|---|---|---|
| Solubility | |||
| DMSO | 48.84 | 100 |
Preparing Stock Solutions for H2L5186303
The following data is based on the product molecular weight 488.45.
Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which all affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 1 mM | 2.05 mL | 10.24 mL | 20.47 mL |
| 5 mM | 0.41 mL | 2.05 mL | 4.09 mL |
| 10 mM | 0.20 mL | 1.02 mL | 2.05 mL |
| 50 mM | 0.04 mL | 0.20 mL | 0.41 mL |
Calculators
Background References
References are publications that support the biological activity of the product. See our Citations tab to view 10 publications citing the usage of this product.
- Fells Structure-based drug design identifies novel LPA3 antagonists. Bioorg.Med.Chem. 2009 PMID: 19800804
- Fells Identification of non-lipid LPA3 antagonists by virtual screening. Bioorg.Med.Chem. 2008 PMID: 18467108
Product Documents for H2L5186303
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for H2L5186303
For research use only
Citations for H2L5186303
Customer Reviews for H2L5186303 (1)
Have you used H2L5186303?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Species: MouseAssay Type: In VivoCell Line/Tissue: primary mammary epitheliumVerified Customer | Posted 11/18/2018Mouse Mammary epithelial cells were used in a standard mammosphere generating assay in a serum-free (B27 supplement), non-adherent condition. LPA or LPA antagonist (H2L5186303) were added to media and the number of spheres were counted after 6 days of culture. Results showed that 10 uM of the antagonist ameliorated most of the LPA-induced suppression, but did not fully neutralize LPA effects. Of two tested LPA inhibitors (products #4878, #5913), this was less effective by a small margin.
There are no reviews that match your criteria.

