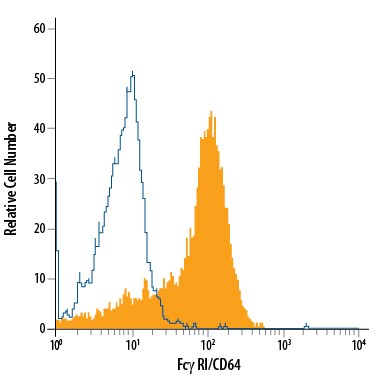
Receptors for the Fc region of IgG (Fc gamma Rs) are members of the Ig superfamily that function in the activation or inhibition of immune responses such as degranulation, phagocytosis, ADCC (antibody-dependent cellular toxicity), cytokine release, and B cell proliferation (1-3). The Fc gamma Rs have been divided into three classes based on close relationships in their extracellular domains; these groups are designated Fc gamma RI (also known as CD64), Fc gamma RII (CD32), and Fc gamma RIII (CD16). Each group may be encoded by multiple genes and exist in different isoforms depending on species and cell type. The CD64 proteins are high affinity receptors (~10‑8 - 10-9 M) capable of binding monomeric IgG, whereas the CD16 and CD32 proteins bind IgG with lower affinities (~10-6 - 10-7 M) only recognizing IgG aggregates surrounding multivalent antigens (1, 4). Fc gamma Rs that deliver an activating signal either have an intrinsic immunoreceptor tyrosine-based activation motif (ITAM) within their cytoplasmic domains or associate with one of the ITAM-bearing adapter subunits, Fc R gamma or zeta (3, 5). The only inhibitory member in human and mouse, Fc gamma RIIb, has an intrinsic cytoplasmic immunoreceptor tyrosine-based inhibitory motif (ITIM). The coordinated functioning of activating and inhibitory receptors is necessary for successful initiation, amplification, and termination of immune responses (5).
Three highly homologous genes (A, B, and C) sharing 98% identity at the nucleotide level have been identified for the human CD64 group (1). Fc gamma RI is transmembrane protein with three extracellular Ig-like domains, and it delivers an activating signal via the associated Fc R gamma accessory chain. The genes for Fc gamma RIB and Fc gamma RIC contain stop codons within their membrane proximal Ig-like domains indicating possible secreted receptors (1, 6). An mRNA splice variant of Fc gamma RIB has a deletion of the membrane-proximal Ig-like domain and encodes a putative transmembrane receptor (6). The high affinity recognition of IgG by Fc gamma RI permits the triggering of effector responses at low IgG concentrations typical of early immune responses (2). Fc gamma RI is expressed constitutively on monocytes and macrophages and can be induced on neutrophils and eosinophils (1, 4). Its expression is up-regulated during bacterial infections and sepsis.