Quinidine
Tocris Bioscience | Catalog # 4108

Key Product Details
Description
Product Description
Quinidine is a class IA antiarrythmic; reduces both Na+ and K+ channel currents, including INa, IKr and IKs. Prolongs QT and induces torsade de pointes (TdP).
Product Specifications for Quinidine
Molecular Weight
Formula
Storage
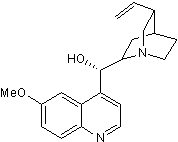
Chemical Name
CAS Number
PubChem ID
InChI Key
SMILES
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Solubility
| Solvent | Max Conc. mg/mL | Max Conc. mM | |
|---|---|---|---|
| Solubility | |||
| DMSO | 32.44 | 100 | |
| Ethanol | 8.11 | 25 |
Preparing Stock Solutions for Quinidine
The following data is based on the product molecular weight 324.42.
Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which all affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 1 mM | 3.08 mL | 15.41 mL | 30.82 mL |
| 5 mM | 0.62 mL | 3.08 mL | 6.16 mL |
| 10 mM | 0.31 mL | 1.54 mL | 3.08 mL |
| 50 mM | 0.06 mL | 0.31 mL | 0.62 mL |
Calculators
Background References
References are publications that support the biological activity of the product. See our Citations tab to view 19 publications citing the usage of this product.
- Borggrefe Short QT syndrome genotype-genotype correlations. J.Electrocardiol. 2005 PMID: 16226079
- Wu Role of late sodium current in modulating the proarrhythmic and antiarrhythmic effects of q.dine Heart Rhythm. 2008 PMID: 19084812
- Caballero Effects of fleca. and q.dine on Kv4.2 currents: voltage dependence and role of S6 valines. Br.J.Pharmacol. 2003 PMID: 12721103
Product Documents for Quinidine
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for Quinidine
For research use only
Related Research Areas
Citations for Quinidine
Customer Reviews for Quinidine (2)
Have you used Quinidine?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
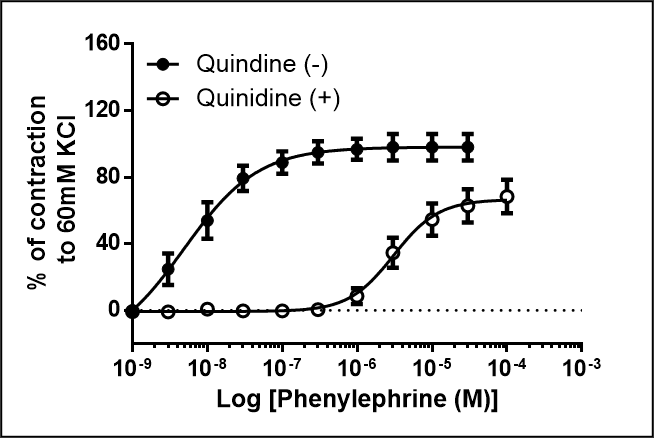
Species: RatAssay Type: In VitroVerified Customer | Posted 12/15/2020Excellent for blocking organic cation transport at 50 uM in rat aorta. However also acts as non-competitive antagonist of alpha-1 adrenoceptors. This is evidenced by the fact it can block vasoconstrictor responses induced by the alpha-1 adrenoceptor agonist phenylephrine in a non-competitive manner.
-
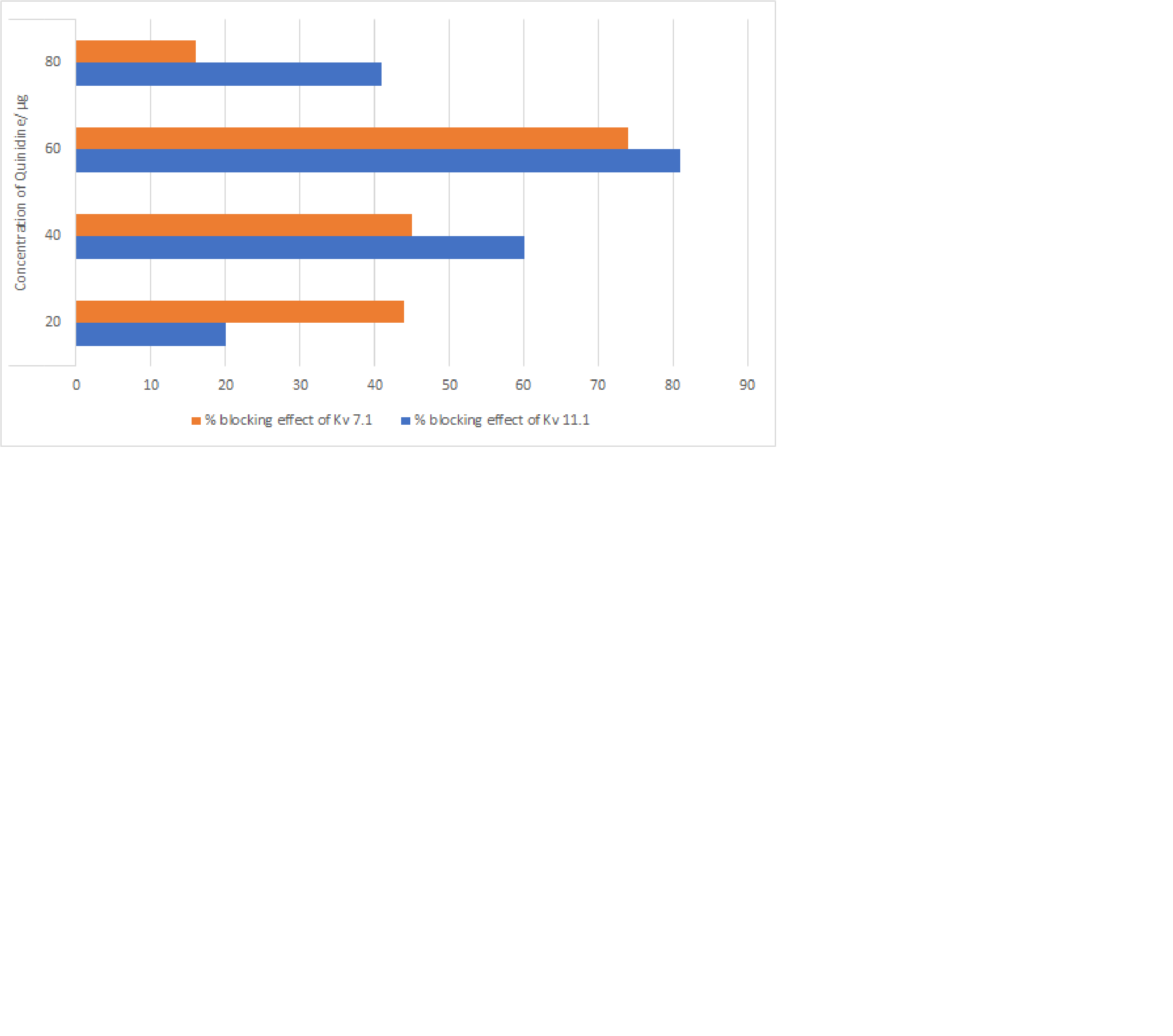
Species: HumanVerified Customer | Posted 12/21/2018Product was used on voltage gated potassium channels (Kv11.1 and Kv7.1) to understand blocking effect.
There are no reviews that match your criteria.