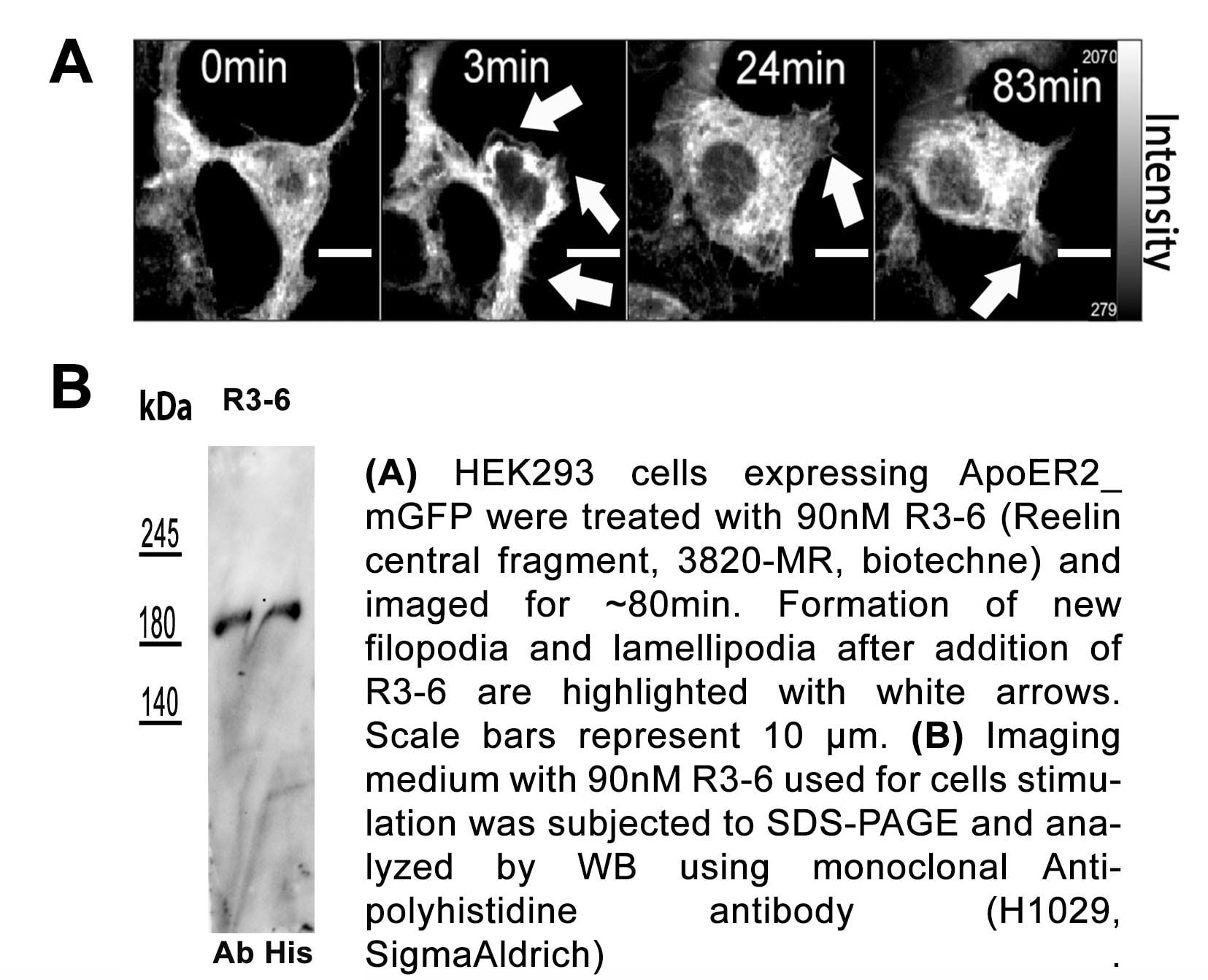
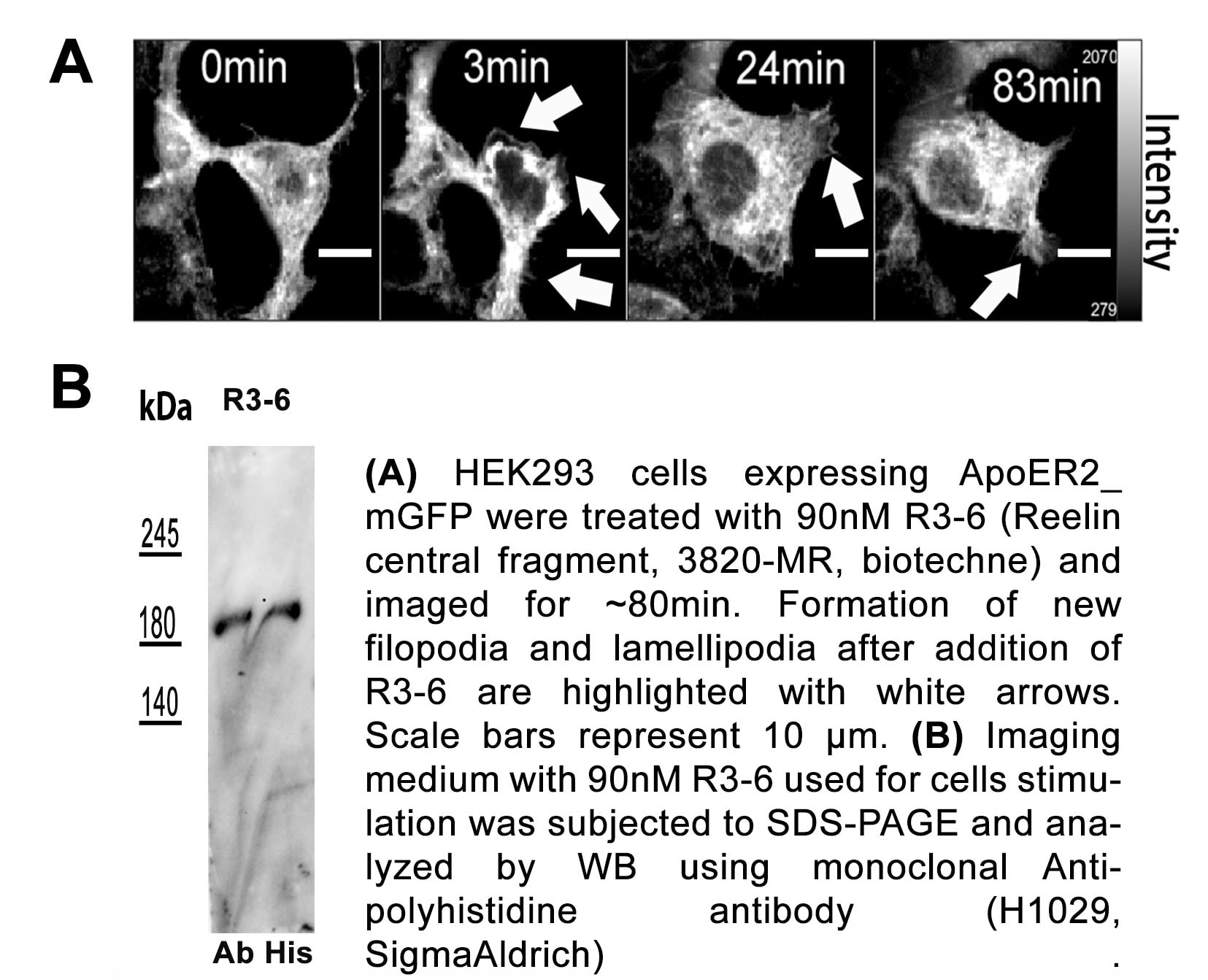
Reelin is secreted by Cajal-Retzius cells in the embryo (1, 4, 11). In the adult, it is expressed in the subventricular zone, rostral migratory stream, olfactory bulb, and in the CA1, CA3, and dentate gyrus regions of the hippocampus, as well as in cerebellar granule cells, pyramidal cells of the entorhinal cortex, GABA interneurons, and glial cells (1, 6, 12, 13). Reelin utilizes the receptors VLDLR and ApoE R2, which have been suggested to have divergent roles in Reelin-mediated neuronal migration (1, 2, 6, 12). It has also been shown to interact with Integrin alpha 3 beta 1 and APP (1, 6, 14, 15). During cortical plate development, Reelin controls cell-cell interactions critical for proper neuronal migration and positioning (1, 2, 4, 5, 11, 12, 16). In the adult, it plays a role in dendrite growth and maturation, and synapse formation (2, 6, 15). Additionally, Reelin has been shown to modulate synaptic transmission and plasticity by regulating the subunit composition and conductivity of NMDA receptors (2, 6, 17). Mutation of the human RELN gene results in lissencephaly with cerebellar hypoplasia (11, 18). In addition, abnormal Reelin expression in human brain has been associated with a variety of cognitive pathological conditions including autism, schizophrenia, bipolar disorder, major depression, and Alzheimer’s disease (1, 6, 11, 13, 19, 20). Reelin deficiency found in Reeler mutant mice causes ataxia, tremors, and impaired motor coordination (4, 16). Peripherally, Reelin is important in the development of neuromuscular junctions. But instead of utilizing the locally expressed ApoE R2 and VLDLR, this function requires the serine protease activity of Reelin (3, 21).
Recombinant Mouse Reelin Protein
R&D Systems | Catalog # 3820-MR


Key Product Details
- R&D Systems NS0-derived Recombinant Mouse Reelin Protein (3820-MR)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
Accession Number
Applications
Product Specifications
Source
Leu1221-Ile2661, with a C-terminal 6-His tag
Purity
Endotoxin Level
N-terminal Sequence Analysis
Predicted Molecular Mass
SDS-PAGE
Activity
Reviewed Applications
Read 2 reviews rated 5 using 3820-MR in the following applications:
Formulation, Preparation, and Storage
Carrier Free
What does CF mean?CF stands for Carrier Free (CF). We typically add Bovine Serum Albumin (BSA) as a carrier protein to our recombinant proteins. Adding a carrier protein enhances protein stability, increases shelf-life, and allows the recombinant protein to be stored at a more dilute concentration. The carrier free version does not contain BSA.
What formulation is right for me?In general, we advise purchasing the recombinant protein with BSA for use in cell or tissue culture, or as an ELISA standard. In contrast, the carrier free protein is recommended for applications, in which the presence of BSA could interfere.
Carrier: 3820-MR
| Formulation | Lyophilized from a 0.2 μm filtered solution in PBS with BSA as a carrier protein. |
| Reconstitution | Reconstitute at 100 μg/mL in sterile PBS containing at least 0.1% human or bovine serum albumin. |
| Shipping | The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Carrier Free: 3820-MR/CF
| Formulation | Lyophilized from a 0.2 μm filtered solution in PBS. |
| Reconstitution | Reconstitute at 100 μg/mL in sterile PBS. |
| Shipping | The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Calculators
Background: Reelin
References
- Fatemi, S.H. (2005) Mol. Psychiatry 10:251.
- Förster, E. et al. (2010) Eur. J. Neurosci. 31:1511.
- Quattrocchi, C.C. et al. (2002) J. Biol. Chem. 277:303.
- D’Arcangelo, G. et al. (1995) Nature 374:719.
- Jossin, Y. et al. (2004) J. Neurosci. 24:514.
- Folsom, T.D. and S.H. Fatemi (2013) Neuropharmacology 68:122.
- Utsunomiya-Tate, N. et al. (2000) Proc. Natl. Acad. Sci. USA 97:9729.
- Nakano, Y. et al. (2007) J. Biol. Chem. 282:20544.
- Hibi, T. et al. (2009) Neurosci. Res. 63:251.
- Lambert de Rouvroit, C. et al. (1999) Exp. Neurol. 156:214.
- Meyer, G. (2010) J. Anat. 217:334.
- D’Arcangelo, G. et al. (1999) Neuron 24:471.
- Senkov, O. et al. (2014) Prog. Brain Res. 214:53.
- Dulabon, L. et al. (2000) Neuron 27:33.
- Hoe, H.S. et al. (2009) J. Neurosci. 29:7459.
- Hirotsune, S. et al. (1995) Nat. Genet. 10:77.
- Levy, A.D. et al. (2014) Front. Neuroanat. 8:116.
- Barros, C.S. et al. (2011) Cold Spring Harb. Perspect. Biol. 3:a005108.
- Botella-López, A. et al. (2006) Proc. Natl. Acad. Sci. USA 103:5573.
- Lubbers, B.R. et al. (2014) Prog. Brain Res. 214:263.
- Quattrocchi, C.C. et al. (2003) Science 301:649.
Alternate Names
Gene Symbol
UniProt
Additional Reelin Products
Product Documents for Recombinant Mouse Reelin Protein
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Mouse Reelin Protein
For research use only
Related Research Areas
Citations for Recombinant Mouse Reelin Protein
Customer Reviews for Recombinant Mouse Reelin Protein (2)
Have you used Recombinant Mouse Reelin Protein?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Application: Cell migration/motilityVerified Customer | Posted 04/09/2019
-
Application: Cell migration/motilityVerified Customer | Posted 04/09/2019
There are no reviews that match your criteria.