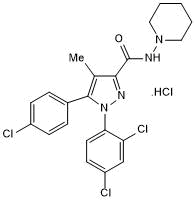
SR 141716A
Tocris Bioscience | Catalog # 0923

Key Product Details
Description
Alternative Names
Product Description
SR 141716A is a potent and selective cannabinoid CB1 receptor antagonist (Ki = 1.98 nM). Also acts as an inverse agonist reversing adenylyl cyclase inhibition by WIN 55,212-2 (Cat.No. 1038) (IC50 = 48 nM). Displays no activity at CB2 receptors. Reduces food intake and body weight in orally dosed non-obese Wistar rats. Also μ-opioid receptor antagonist (Ki = 652 nm)
Product Specifications for SR 141716A
Molecular Weight
Formula
Storage
Purity
Chemical Name
CAS Number
PubChem ID
InChI Key
SMILES
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Solubility
| Solvent | Max Conc. mg/mL | Max Conc. mM | |
|---|---|---|---|
| Solubility | |||
| DMSO | 50.02 | 100 |
Preparing Stock Solutions for SR 141716A
The following data is based on the product molecular weight 500.25.
Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which all affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 1 mM | 2.00 mL | 10.00 mL | 19.99 mL |
| 5 mM | 0.40 mL | 2.00 mL | 4.00 mL |
| 10 mM | 0.20 mL | 1.00 mL | 2.00 mL |
| 50 mM | 0.04 mL | 0.20 mL | 0.40 mL |
Calculators
Background References
References are publications that support the biological activity of the product. See our Citations tab to view 143 publications citing the usage of this product.
- Seely AM-251 and rimon. act as direct antagonists at mu-opioid receptors: implications for opioid/cannabinoid interaction studies. Neuropharmacology 2012 PMID: 22771770
- Colombo Appetite suppression and weight loss after the cannabinoid antagonist SR 141716. Life Sci. 1998 PMID: 9718088
- Rinaldi-Carmona SR 141716A, a potent and selective antagonist of the brain cannabinoid receptor. FEBS Lett. 1994 PMID: 8070571
- Rinaldi-Carmona Biochemical and pharmacological characterisation of SR 141716A, the first potent and selective brain cannabinoid receptor antagonist. Life Sci. 1995 PMID: 7776817
- Janiak Blockade of cannabinoid CB1 receptors improves renal function, metabolic profile, and increased survival of obese Zucker rats. Kidney Int. 2007 PMID: 17882151
Product Documents for SR 141716A
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for SR 141716A
For research use only
Citations for SR 141716A
Customer Reviews for SR 141716A (2)
Have you used SR 141716A?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Species: MouseAssay Type: In VivoVerified Customer | Posted 05/24/2023Rimonabant stock was prepared in dmso and tween80, and consequently freshly diluted with saline.10mg/kg body weight, dissolved in solution with 6.7% dmso, 1.7% tween80 and 91.6% saline; intraperitoneally injected.
-
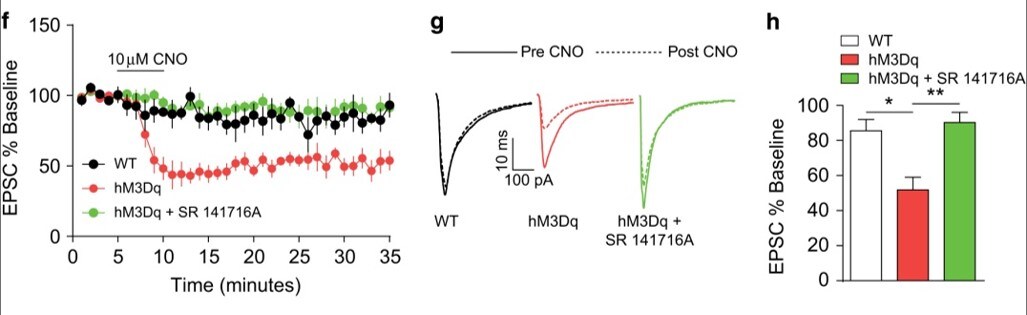
Species: MouseAssay Type: Ex VivoCell Line/Tissue: Bed nucleus of the stria terminalisVerified Customer | Posted 06/01/2020SR 141716A was solubilized in DMSO and bath applied at a 5 micromolar concentration. It effectively blocked CB1-receptor-dependent long term depression of evoked excitatory postsynaptic current in the bed nucleus of the stria terminalis.
There are no reviews that match your criteria.