Having trouble trying to find the right product? Chat with our scientists below - we're here to help!
141 results for "SOD3/EC-SOD" in Products
SOD3/EC-SOD Products
Superoxide Dismutases, originally identified as Indophenoloxidases (IPOs), are enzymes that catalyze the conversion of naturally-occuring but harmful superoxide radicals into molecular oxygen and hydrogen peroxide. Three mammalian isozymes of SOD have been identified and are functionally related but have very modest sequence homology. SODs are typically soluble secreted or cytosolic proteins, but are also found in the mitochondria and extracellular matrix.
Any of three metals, manganese, ...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | WB, Simple Western |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB, Simple Western |
Loading...
| Reactivity: | Human, Mouse, Rat, Guinea Pig |
| Details: | Mouse IgG1 kappa Monoclonal Clone #4GG11G6 |
| Applications: | IHC, WB, ELISA, ICC/IF |
Loading...
| Reactivity: | Human, Mouse |
| Details: | Mouse IgG1 Monoclonal Clone #713707 |
| Applications: | WB, Simple Western |
Loading...
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Human |
| Source: | Wheat germ |
| Applications: | WB, ELISA, AP, MA, PAGE |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Human, Mouse |
| Details: | Mouse IgG1 Monoclonal Clone #713707 |
| Applications: | WB |
Loading...
| Reactivity: | Human, Mouse |
| Details: | Mouse IgG1 Monoclonal Clone #713707 |
| Applications: | WB |
Loading...
| Reactivity: | Human, Mouse |
| Details: | Mouse IgG1 Monoclonal Clone #713707 |
| Applications: | WB |
Loading...
| Reactivity: | Human, Mouse |
| Details: | Mouse IgG1 Monoclonal Clone #713707 |
| Applications: | WB |
Loading...
| Reactivity: | Human, Mouse |
| Details: | Mouse IgG1 Monoclonal Clone #713707 |
| Applications: | WB |
Loading...
| Reactivity: | Human, Mouse |
| Details: | Mouse IgG1 Monoclonal Clone #713707 |
| Applications: | WB |
Loading...
| Reactivity: | Human, Mouse |
| Details: | Mouse IgG1 Monoclonal Clone #713707 |
| Applications: | WB |
Loading...
| Reactivity: | Human, Mouse |
| Details: | Mouse IgG1 Monoclonal Clone #713707 |
| Applications: | WB |
Loading...
| Reactivity: | Mouse |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
Recombinant Monoclonal Antibody
| Reactivity: | Human |
| Details: | Rabbit IgG Monoclonal Clone #SR1423 |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | WB |
Loading...



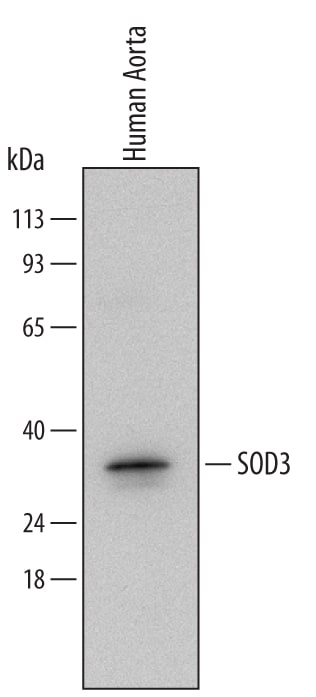

![Western Blot: SOD3/EC-SOD Antibody (4GG11G6) [NBP1-22417] Western Blot: SOD3/EC-SOD Antibody (4GG11G6) [NBP1-22417]](https://resources.rndsystems.com/images/products/SOD3-EC-SOD-Antibody-4GG11G6-Western-Blot-NBP1-22417-img0004.jpg)

![Western Blot: SOD3/EC-SOD Antibody [NBP2-38493] Western Blot: SOD3/EC-SOD Antibody [NBP2-38493]](https://resources.rndsystems.com/images/products/SOD3-EC-SOD-Antibody-Western-Blot-NBP2-38493-img0002.jpg)
![SDS-PAGE: Recombinant Human SOD3/EC-SOD GST (N-Term) Protein [H00006649-Q01] SDS-PAGE: Recombinant Human SOD3/EC-SOD GST (N-Term) Protein [H00006649-Q01]](https://resources.rndsystems.com/images/products/SOD3-EC-SOD-Partial-Recombinant-Protein-SDS-Page-H00006649-Q01-img0001.jpg)
![Western Blot: SOD3/EC-SOD Antibody (SR1423) [NBP3-21798] -](https://resources.rndsystems.com/images/products/nbp3-21798_rabbit-sod3-ec-sod-mab-sr1423-6720239533729.jpg)