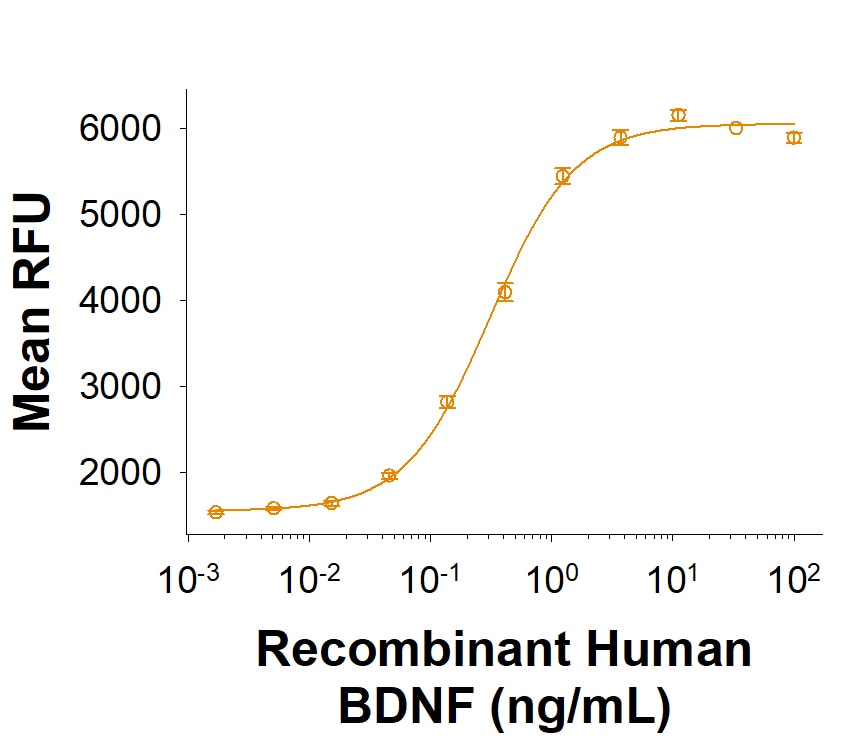
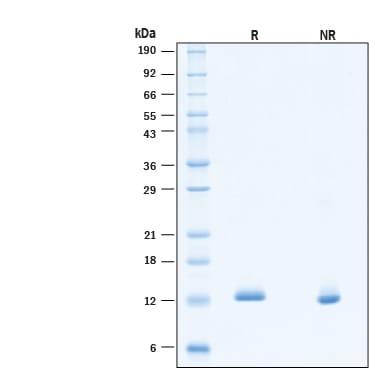
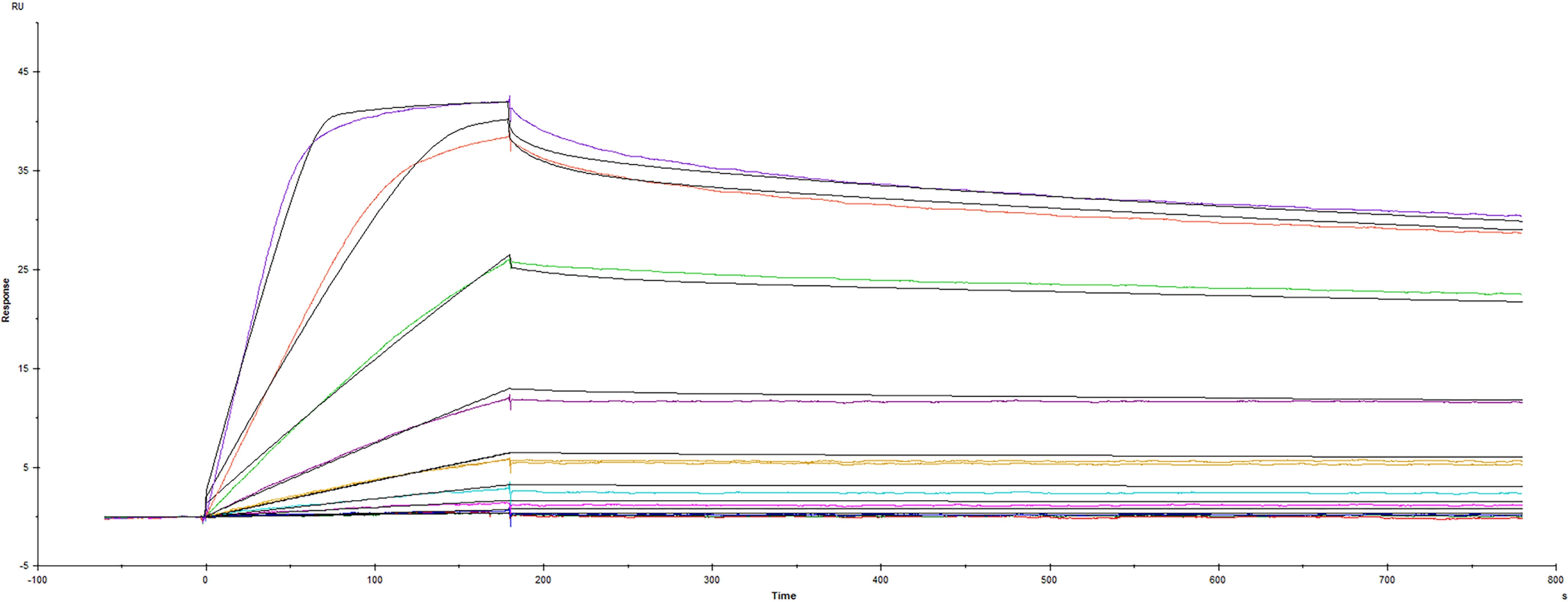
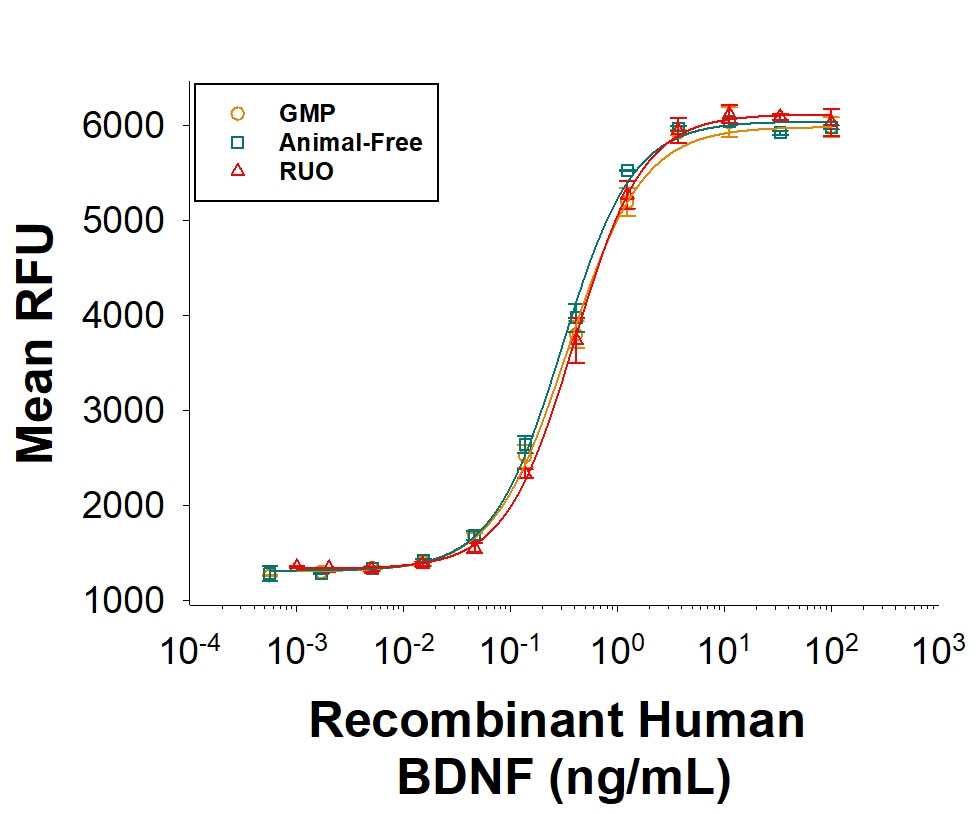
BDNF
is often utilized as a cell culture supplement to promote neural
differentiation in regenerative medicine studies and clinical manufacturing
protocols.
The
neurotrophin family is comprised of at least four proteins including BDNF,
nerve growth factor (NGF), neurotrophin-3 (NT3), and NT4/5. Human BDNF is
initially expressed as a proprotein, which is then cleaved to yield a mature
protein. Mature BDNF is a non-covalently linked homodimer, with each monomer
containing antiparallel beta -strands and a characteristic cystine knot motif.
Within the mature domain, human BDNF shares the identical amino acid sequence
with mature mouse and rat BDNF.
BDNF
is strongly expressed in various regions of the brain, including the
hippocampus and cerebellum, and weaker expression has been detected in the
thymus, liver, spleen, heart, and lung. BDNF participates in axonal growth and
pathfinding and in the modulation of dendritic growth and morphology and in
later stages of development regulates synaptic transmission and plasticity and
acts as a central modulator of pain. BDNF binds with high affinity and
specifically activates the TrkB tyrosine kinase receptor. BDNF signaling via
TrkB is essential for adult synaptic plasticity and the formation of memories.
The
BDNF signaling pathway utilizes both AKT and ERK pathways to exert its pleiotrophic
effects in the central nervous system. Decreased expression of BDNF is seen in
many neurological diseases such as Alzheimer's disease, Parkinson's disease,
Huntington's disease, and autism. Further, BDNF is proposed as a biomarker for
psychiatric disorders such as bipolar disease and depression and has been
implicated in posttraumatic stress disorder, phobia, and panic disorder. A
single amino acid substitution, Val66Met, has been shown to lead to reduced,
activity-dependent BDNF secretion and memory impairment.