Recombinant Human IL-8/CXCL8 Protein
R&D Systems | Catalog # 208-IL

Key Product Details
- R&D Systems E. coli-derived Recombinant Human IL-8/CXCL8 Protein (208-IL)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
Accession Number
Applications
Product Specifications
Source
Ser28-Ser99
Purity
Endotoxin Level
N-terminal Sequence Analysis
Predicted Molecular Mass
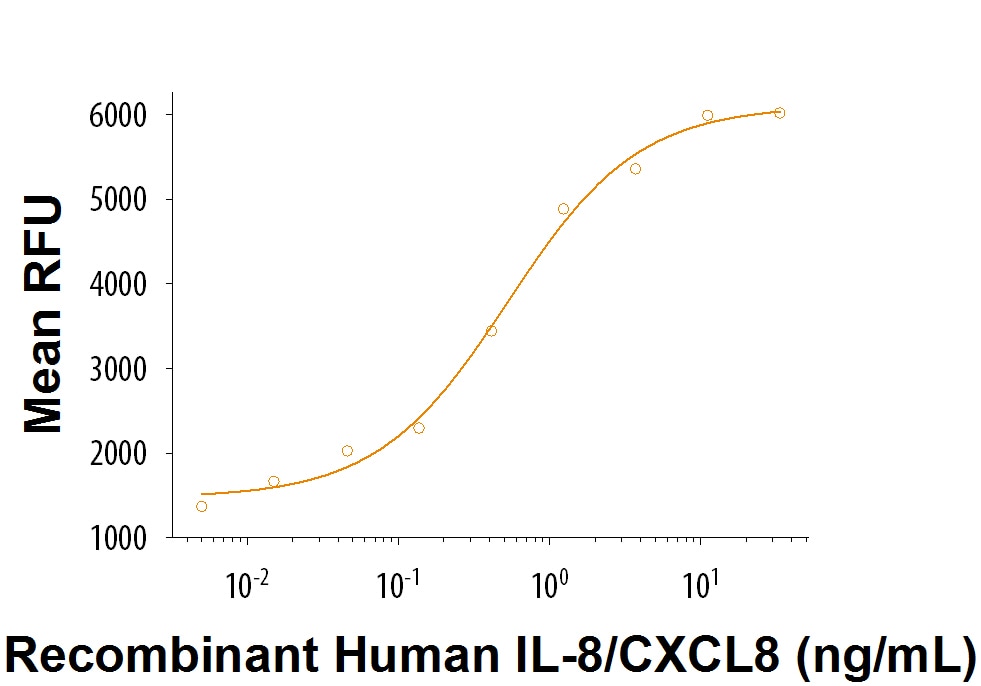
Activity
The ED50 for this effect is 0.5-2.5 ng/mL.
Reviewed Applications
Read 8 reviews rated 4.9 using 208-IL in the following applications:
Scientific Data Images for Recombinant Human IL-8/CXCL8 Protein
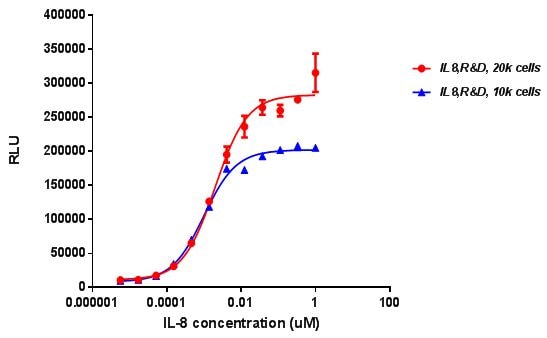
Recombinant Human IL-8/CXCL8 Protein Bioactivity
Recombinant Human IL-8/CXCL8 (Catalog # 208-IL) chemoattracts the BaF3 mouse pro-B cell line transfected with human CXCR2. The ED50 for this effect is 0.5-2.5 ng/mL.Recombinant Human IL-8/CXCL8 Protein SDS-PAGE
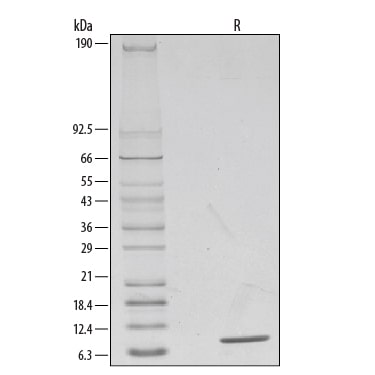
1 μg/lane of Recombinant Human IL-8/CXCL8 was resolved with SDS-PAGE under reducing (R) conditions and visualized by silver staining, showing a single band at 8 kDa.Formulation, Preparation, and Storage
Carrier Free
What does CF mean?CF stands for Carrier Free (CF). We typically add Bovine Serum Albumin (BSA) as a carrier protein to our recombinant proteins. Adding a carrier protein enhances protein stability, increases shelf-life, and allows the recombinant protein to be stored at a more dilute concentration. The carrier free version does not contain BSA.
What formulation is right for me?In general, we advise purchasing the recombinant protein with BSA for use in cell or tissue culture, or as an ELISA standard. In contrast, the carrier free protein is recommended for applications, in which the presence of BSA could interfere.
Carrier: 208-IL
| Formulation | Lyophilized from a 0.2 μm filtered solution in PBS with BSA as a carrier protein. |
| Reconstitution | Reconstitute at 100 μg/mL in sterile PBS containing at least 0.1% human or bovine serum albumin. |
| Shipping | The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Carrier Free: 208-IL/CF
| Formulation | Lyophilized from a 0.2 μm filtered solution in PBS. |
| Reconstitution | Reconstitute at 100 μg/mL in sterile PBS. |
| Shipping | The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Calculators
Background: IL-8/CXCL8
References
- Lazennec, G. and A. Richmond (2010) Trends Mol. Med. 16:133.
- Nesmelova, I.V. et al. (2005) J. Biol. Chem. 280:4948.
- Pichert, A. et al. (2012) Biomatter 2:142.
- Schmid, J. and C. Weissmann (1987) J. Immunol. 139:250.
- Mortier, A. et al. (2008) Pharmacol. Ther. 120:197.
- Proost, P. et al. (2008) J. Exp. Med. 205:2085.
- Neote, K. et al. (1994) Blood 84:44.
- Munoz, L.M. et al. (2009) J. Immunol. 183:7337.
- Gerszten, R.E. et al. (1999) Nature 398:718.
- Jones, S.A. et al. (1996) Proc. Natl. Acad. Sci. USA 93:6682.
- Bergin, D.A. et al. (2010) J. Clin. Invest. 120:4236.
- Apostolakis, S. et al. (2009) Cardiovasc. Res. 84:353.
- Martin, D. et al. (2009) J. Biol. Chem. 284:6038.
- Li, A. et al. (2005) Angiogenesis 8:63.
- Waugh, D.J. and C. Wilson (2008) Clin. Cancer Res. 14:6735.
- Fernando, R.I. et al. (2011) Cancer Res. 71:5296.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional IL-8/CXCL8 Products
Product Documents for Recombinant Human IL-8/CXCL8 Protein
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Human IL-8/CXCL8 Protein
For research use only
Citations for Recombinant Human IL-8/CXCL8 Protein
Customer Reviews for Recombinant Human IL-8/CXCL8 Protein (8)
Have you used Recombinant Human IL-8/CXCL8 Protein?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
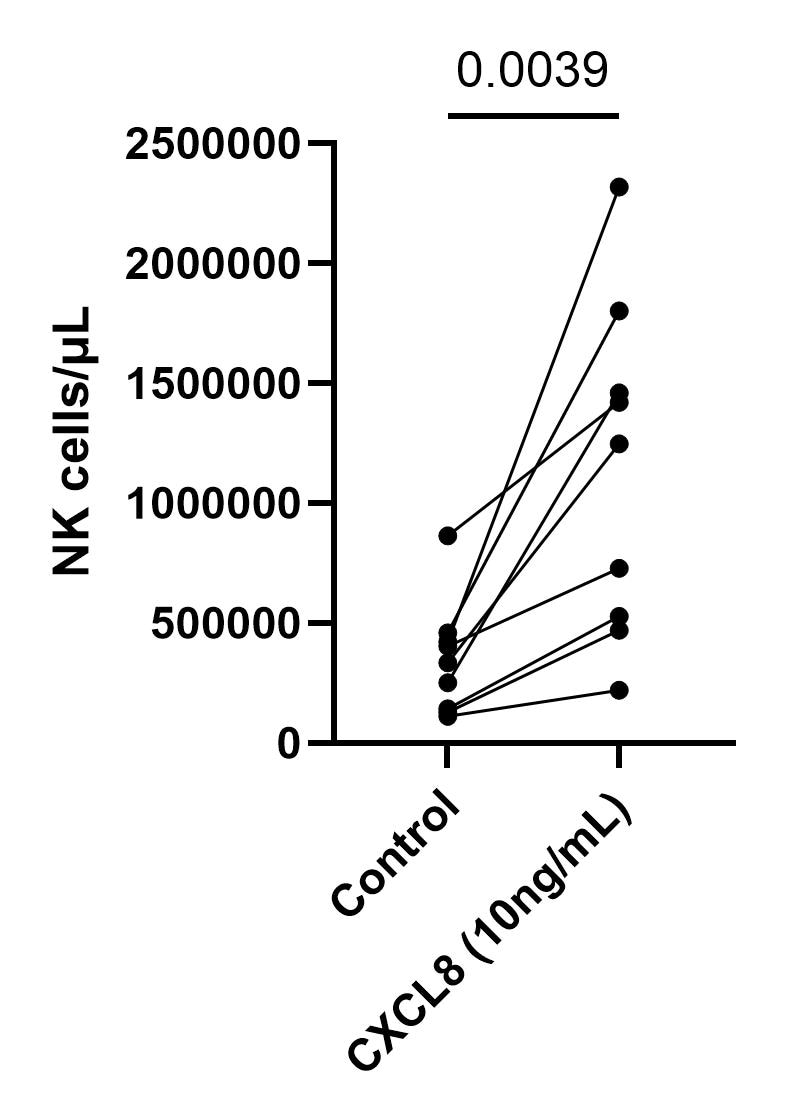
Application: Cell migration/motilitySpecies: HumanVerified Customer | Posted 04/24/2025Comparison of NK cell count/µL in the control condition (R10) vs CXCL8 at 10ng/mL200.000 sorted NK cells were place in the upper chamber of a 24 well plate with transwell inserts (5µm pore size). 650µL of culture media containing 10ng/mL of CXCL8 were placed in the lower chamber. Cell count was performed by flow cytometry using counting bead after one hour of migration at 37ºC.
-
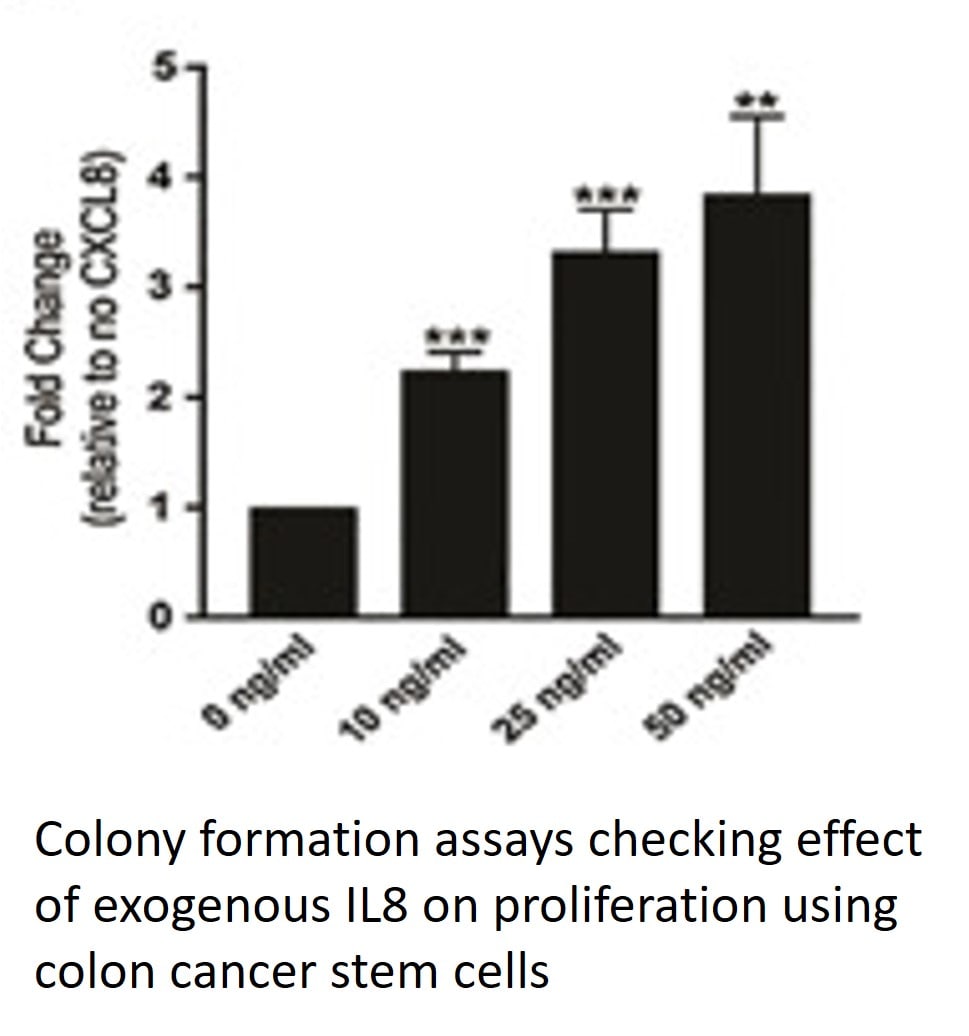
Application: Cell ProliferationVerified Customer | Posted 08/14/2023
-
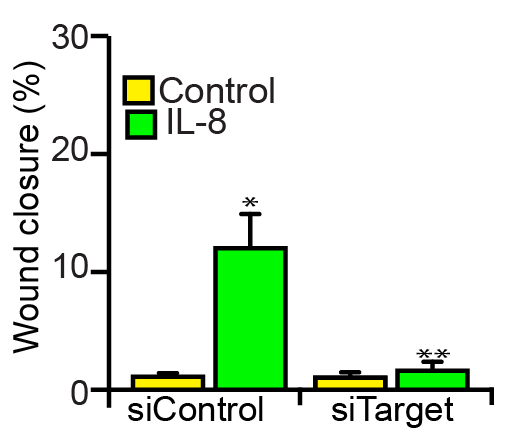
Application: Cell migration/motilityVerified Customer | Posted 03/15/2021The primary cells were starved overnight and treated with IL-8 (40ng/ml) and tested its effects on cell migration.
-
Application: Assay validationVerified Customer | Posted 02/09/2021
-
Application: In vitro bioactivity in cell cultureVerified Customer | Posted 12/03/2018rhIL-8 induce beta -arrestin release in CXCR1/U2OS reporter cell line
-
Application: Immunoassay StandardVerified Customer | Posted 11/26/2018The recombinant human IL-8 protein was used as the calibration reagent in MSD assay with biotinylated MAB208 (capture) and Sulfo-Tag labeled AF-208-NA (detection) antibodies.
-
Application: activation of neutrophils from blood in vitro.Verified Customer | Posted 11/10/2017
-
Application: activation of neutrophilsVerified Customer | Posted 04/07/2017Neutrophils were activated with 208-IL and assessed for morphological changes and migratory activity on 2D cell culture plate.
There are no reviews that match your criteria.
FAQs for Recombinant Human IL-8/CXCL8 Protein
-
Q: Are mouse or rat CXCL8/IL-8 products offered?
A: Mouse and rat do not have a gene which encodes CXCL8/IL-8. In mouse, the functional homologs to human CXCL8/IL-8 are CXCL1/KC and CXCL2/MIP-2. In rat, the functional homolog to CXCL8/human IL-8 is CXCL3/CINC-2.
-
Q: Can Recombinant Human IL-8/CXCL8 Protein (Catalog # 208-IL) be used with mouse cells?
A: Bioactivity of Recombinant Human IL-8/CXCL8 Protein (Catalog # 208-IL) has only been evaluated using human cells transfected with human CXCR2 (the human receptor for IL-8), and we have not evaluated the activity of this protein using mouse cells. There is no IL-8/CXCL8 gene counterpart in rodent species. We are aware of a few publications citing use of 208-IL in mouse assay systems, so we would recommend reviewing the publications listed on the Citations tab of the product-specific web page to find reported use of our products in similar experimental layouts.
-
Q: What is the difference between 208-IL (rhCXCL8/IL-8) and 618-IL (rhEndothelial Cell-Derived IL-8/CXCL8)?
A: Several N-terminal processed forms of CXCL8/IL-8 are produced by proteolytic cleavage after secretion by cells such as peripheral blood monocytes, leukcocytes and endothelial cells. Catalog # 208-IL consists only of the sequence Ser28 to Ser99 (also known as IL-8, 6-77), which is the most prominent form of IL-8. According to literature, IL-8(6-77) has a 5-10-fold higher activity in neutrophil activation. Catalog # 618-IL consists of Ser28 to Ser99 (IL-8, 6-77) and Ala23 to Ser99 (IL-8). In our chemoattraction assay, 208-IL has a 2-fold higher activity than 618-IL.Several N-terminal processed forms of CXCL8/IL-8 are produced by proteolytic cleavage after secretion by cells such as peripheral blood monocytes, leukcocytes and endothelial cells. Catalog # 208-IL consists only of the sequence Ser28 to Ser99 (also known as IL-8, 6-77), which is the most prominent form of IL-8. According to literature, IL-8(6-77) has a 5-10-fold higher activity in neutrophil activation. Catalog # 618-IL consists of Ser28 to Ser99 (IL-8, 6-77) and Ala23 to Ser99 (IL-8). In our chemoattraction assay, 208-IL has a 2-fold higher activity than 618-IL.
-
Q: Are mouse or rat CXCL8/IL-8 products offered?
A: Mouse and rat do not have a gene which encodes CXCL8/IL-8. In mouse, the functional homologs to human CXCL8/IL-8 are CXCL1/KC and CXCL2/MIP-2. In rat, the functional homolog to CXCL8/human IL-8 is CXCL3/CINC-2.
-
Q: Can Recombinant Human IL-8/CXCL8 Protein (Catalog # 208-IL) be used with mouse cells?
A: Bioactivity of Recombinant Human IL-8/CXCL8 Protein (Catalog # 208-IL) has only been evaluated using human cells transfected with human CXCR2 (the human receptor for IL-8), and we have not evaluated the activity of this protein using mouse cells. There is no IL-8/CXCL8 gene counterpart in rodent species. We are aware of a few publications citing use of 208-IL in mouse assay systems, so we would recommend reviewing the publications listed on the Citations tab of the product-specific web page to find reported use of our products in similar experimental layouts.
-
Q: What is the difference between 208-IL (rhCXCL8/IL-8) and 618-IL (rhEndothelial Cell-Derived IL-8/CXCL8)?
A: Several N-terminal processed forms of CXCL8/IL-8 are produced by proteolytic cleavage after secretion by cells such as peripheral blood monocytes, leukcocytes and endothelial cells. Catalog # 208-IL consists only of the sequence Ser28 to Ser99 (also known as IL-8, 6-77), which is the most prominent form of IL-8. According to literature, IL-8(6-77) has a 5-10-fold higher activity in neutrophil activation. Catalog # 618-IL consists of Ser28 to Ser99 (IL-8, 6-77) and Ala23 to Ser99 (IL-8). In our chemoattraction assay, 208-IL has a 2-fold higher activity than 618-IL.Several N-terminal processed forms of CXCL8/IL-8 are produced by proteolytic cleavage after secretion by cells such as peripheral blood monocytes, leukcocytes and endothelial cells. Catalog # 208-IL consists only of the sequence Ser28 to Ser99 (also known as IL-8, 6-77), which is the most prominent form of IL-8. According to literature, IL-8(6-77) has a 5-10-fold higher activity in neutrophil activation. Catalog # 618-IL consists of Ser28 to Ser99 (IL-8, 6-77) and Ala23 to Ser99 (IL-8). In our chemoattraction assay, 208-IL has a 2-fold higher activity than 618-IL.
-
Q: Are mouse or rat CXCL8/IL-8 products offered?
A: Mouse and rat do not have a gene which encodes CXCL8/IL-8. In mouse, the functional homologs to human CXCL8/IL-8 are CXCL1/KC and CXCL2/MIP-2. In rat, the functional homolog to CXCL8/human IL-8 is CXCL3/CINC-2.
-
Q: Can Recombinant Human IL-8/CXCL8 Protein (Catalog # 208-IL) be used with mouse cells?
A: Bioactivity of Recombinant Human IL-8/CXCL8 Protein (Catalog # 208-IL) has only been evaluated using human cells transfected with human CXCR2 (the human receptor for IL-8), and we have not evaluated the activity of this protein using mouse cells. There is no IL-8/CXCL8 gene counterpart in rodent species. We are aware of a few publications citing use of 208-IL in mouse assay systems, so we would recommend reviewing the publications listed on the Citations tab of the product-specific web page to find reported use of our products in similar experimental layouts.
-
Q: What is the difference between 208-IL (rhCXCL8/IL-8) and 618-IL (rhEndothelial Cell-Derived IL-8/CXCL8)?
A: Several N-terminal processed forms of CXCL8/IL-8 are produced by proteolytic cleavage after secretion by cells such as peripheral blood monocytes, leukcocytes and endothelial cells. Catalog # 208-IL consists only of the sequence Ser28 to Ser99 (also known as IL-8, 6-77), which is the most prominent form of IL-8. According to literature, IL-8(6-77) has a 5-10-fold higher activity in neutrophil activation. Catalog # 618-IL consists of Ser28 to Ser99 (IL-8, 6-77) and Ala23 to Ser99 (IL-8). In our chemoattraction assay, 208-IL has a 2-fold higher activity than 618-IL.Several N-terminal processed forms of CXCL8/IL-8 are produced by proteolytic cleavage after secretion by cells such as peripheral blood monocytes, leukcocytes and endothelial cells. Catalog # 208-IL consists only of the sequence Ser28 to Ser99 (also known as IL-8, 6-77), which is the most prominent form of IL-8. According to literature, IL-8(6-77) has a 5-10-fold higher activity in neutrophil activation. Catalog # 618-IL consists of Ser28 to Ser99 (IL-8, 6-77) and Ala23 to Ser99 (IL-8). In our chemoattraction assay, 208-IL has a 2-fold higher activity than 618-IL.