109 results for "GRB2" in 产品
GRB2 Products
GRB-2 (growth factor receptor-bound protein 2), an adaptor protein involved in signal transduction, contains a central SH2 domain flanked by two SH3 domains. The SH2 domain binds to phosphotyrosine residues in RTKs such as PDGF and EGF, non-RTKs such as Bcr/Abl and FAK, and docking proteins such as FRS-2 and Gab1. The SH3 domains associate with proline rich motifs on the guanine nucleotide releasing factor, Sos, stimulating GTP binding to Ras, which in turn activates MAPK and other signaling pathways. GRB-2 also participates in the endocytosis of EGFR through its recruitment of Cbl. Tyrosine phosphorylation of GRB-2 SH3 domains reduces binding to Sos and negatively regulates downstream signaling pathways including Ras, JNK and MAPK.
| Reactivity: | Human, Mouse, Rat |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
| Reactivity: | Human, Mouse, Rat, Porcine |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB, ELISA |
Recombinant Monoclonal Antibody
| Reactivity: | Human, Mouse, Rat |
| Details: | Rabbit IgG Monoclonal Clone #9K6H0 |
| Applications: | IHC, WB, ELISA, ICC/IF, IP |
| Reactivity: | Human |
| Details: | Mouse IgG2B Monoclonal Clone #669604 |
| Applications: | IHC, WB |
| Reactivity: | Human |
| Source: | Wheat germ |
| Applications: | WB, ELISA, AP, MA |
| Reactivity: | Human, Mouse, Rat |
| Details: | Mouse IgG2B Monoclonal Clone #481021 |
| Applications: | WB |
Recombinant Monoclonal Antibody
| Reactivity: | Human, Mouse, Rat, Monkey |
| Details: | Rabbit IgG Monoclonal Clone #JE49-73 |
| Applications: | WB, Flow, ICC/IF |
Recombinant Monoclonal Antibody
| Reactivity: | Human, Mouse, Rat |
| Details: | Rabbit IgG Monoclonal Clone #6E1 |
| Applications: | IHC, WB, ELISA |
Recombinant Monoclonal Antibody
| Reactivity: | Human, Mouse, Rat |
| Details: | Rabbit IgG Monoclonal Clone #SR1396 |
| Applications: | IHC, WB, ICC/IF, IP |
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | WB, ICC/IF |
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | WB, ICC/IF |
| Reactivity: | Human, Mouse, Rat |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
| Reactivity: | Human, Mouse, Rat |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
| Reactivity: | Human, Mouse, Rat |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
| Reactivity: | Human, Mouse, Rat |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
| Reactivity: | Human, Mouse, Rat |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
| Reactivity: | Human, Mouse, Rat |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
| Reactivity: | Human, Mouse, Rat |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
| Reactivity: | Human, Mouse, Rat |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
| Reactivity: | Human |
| Details: | Mouse IgG2B Monoclonal Clone #669604 |
| Applications: | IHC, WB |
| Reactivity: | Human |
| Details: | Mouse IgG2B Monoclonal Clone #669604 |
| Applications: | IHC, WB |
| Reactivity: | Human |
| Details: | Mouse IgG2B Monoclonal Clone #669604 |
| Applications: | IHC, WB |
| Reactivity: | Human |
| Details: | Mouse IgG2B Monoclonal Clone #669604 |
| Applications: | IHC, WB |
| Reactivity: | Human |
| Details: | Mouse IgG2B Monoclonal Clone #669604 |
| Applications: | IHC, WB |
| Reactivity: | Human |
| Details: | Mouse IgG2B Monoclonal Clone #669604 |
| Applications: | IHC, WB |




![Western Blot: GRB2 Antibody [NB100-866] Western Blot: GRB2 Antibody [NB100-866]](https://resources.rndsystems.com/images/products/GRB2-Antibody-Western-Blot-NB100-866-img0012.jpg)
![Western Blot: GRB2 Antibody (9K6H0) [NBP3-15638] Western Blot: GRB2 Antibody (9K6H0) [NBP3-15638]](https://resources.rndsystems.com/images/products/GRB2-Antibody-9K6H0-Western-Blot-NBP3-15638-img0005.jpg)
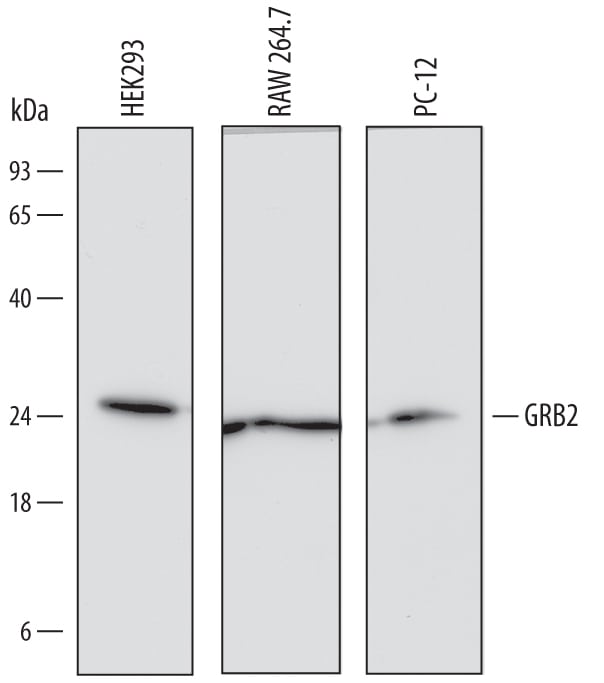
![Western Blot: Recombinant Human GRB2 GST (N-Term) Protein [H00002885-P01] Western Blot: Recombinant Human GRB2 GST (N-Term) Protein [H00002885-P01]](https://resources.rndsystems.com/images/products/Recombinant-Human-GRB2-Protein-Western-Blot-H00002885-P01-img0003.jpg)

![Western Blot: GRB2 Antibody (JE49-73) [NBP3-32403] GRB2 Antibody (JE49-73)](https://resources.rndsystems.com/images/products/nbp3-32403_rabbit-grb2-mab-je49-73-278202415295585.jpg)
![Western Blot: GRB2 Antibody (6E1) [NBP3-15167] Western Blot: GRB2 Antibody (6E1) [NBP3-15167]](https://resources.rndsystems.com/images/products/GRB2-Antibody-6E1-Western-Blot-NBP3-15167-img0002.jpg)
![Western Blot: GRB2 Antibody (SR1396) [NBP3-21851] -](https://resources.rndsystems.com/images/products/nbp3-21851_rabbit-grb2-mab-sr1396-57202315545124.jpg)
![Western Blot: GRB2 Antibody [NBP2-55208] Western Blot: GRB2 Antibody [NBP2-55208]](https://resources.rndsystems.com/images/products/GRB2-Antibody-Western-Blot-NBP2-55208-img0003.jpg)
![Western Blot: GRB2 Antibody [NBP2-55209] Western Blot: GRB2 Antibody [NBP2-55209]](https://resources.rndsystems.com/images/products/GRB2-Antibody-Western-Blot-NBP2-55209-img0003.jpg)