Having trouble trying to find the right product? Chat with our scientists below - we're here to help!
148 results for "PKC epsilon" in 产品
PKC epsilon Products
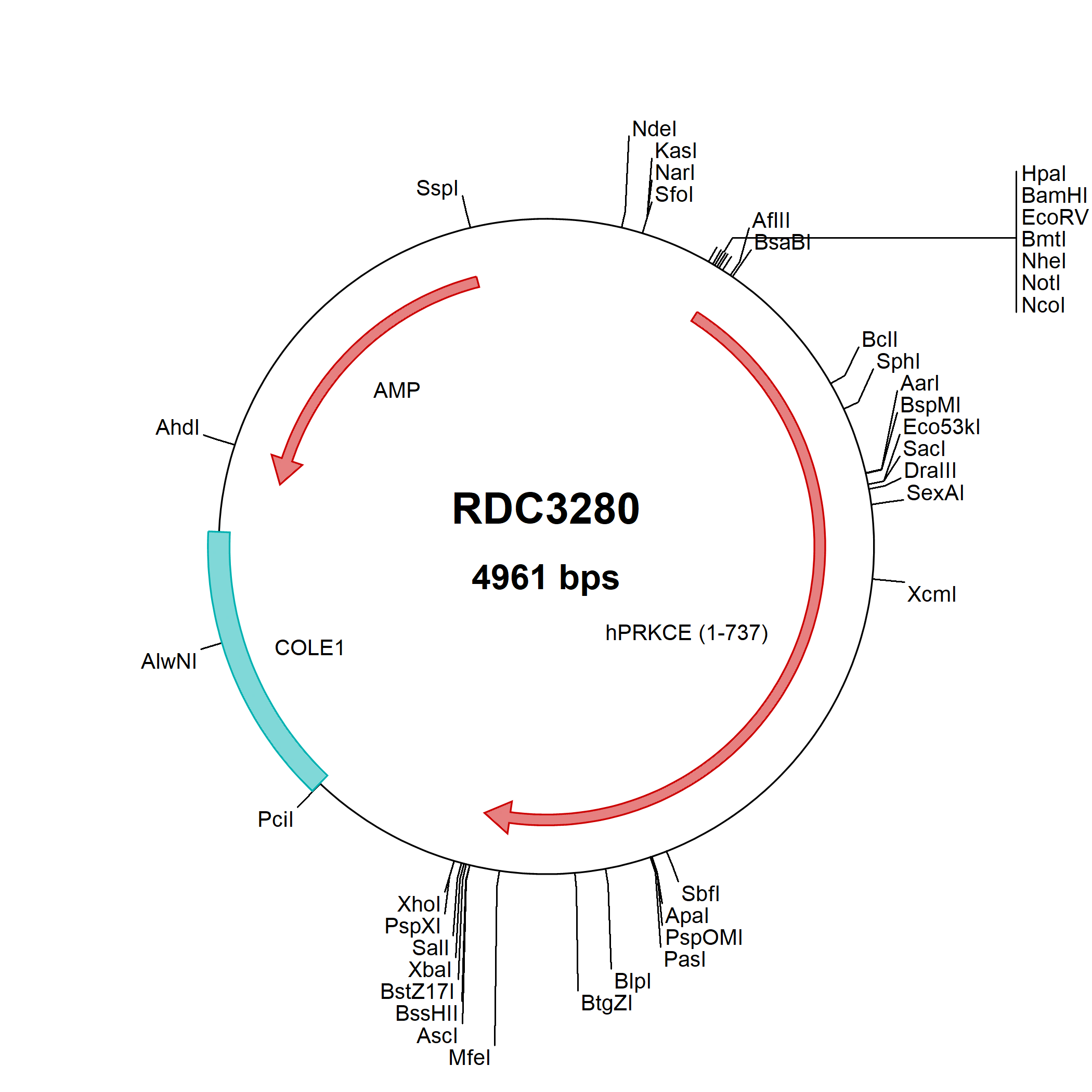
PKC epsilon (protein kinase C-epsilon) is an 87 kDa member of the novel PKC subfamily, AGC Ser/Thr protein kinase family of enzymes. It is a widely-expressed Ca++-insensitive, phospholipid-dependent enzyme that catalyzes the phosphorylation of multiple proteins. Human PKC epsilon is 737 amino acids (aa) in length. It contains two general regions: a non-Ca++-binding plus lipid binding regulatory region (aa 1-99 and 169-292, respectively), and an ATP-binding catalytic domain (aa 408-668). ...
Protein kinase C inhibitor
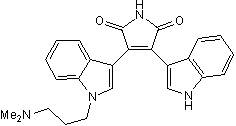
| Alternate Names: | Gö 6850,Bisindolylmaleimide I |
| Chemical Name: | 2-[1-(3-Dimethylaminopropyl)indol-3-yl]-3-(indol-3-yl) maleimide |
| Purity: | ≥98% |
Loading...
| Reactivity: | Human, Mouse, Rat |
| Details: | Sheep IgG Polyclonal |
| Applications: | IHC, WB, Simple Western |
Loading...
| Reactivity: | Human, Mouse, Rat, Monkey, Primate |
| Details: | Mouse IgG2B Monoclonal Clone #OTI4G8 |
| Applications: | IHC, WB, Flow, ICC/IF |
Loading...
| Reactivity: | Human, Mouse, Rat, Monkey, Primate |
| Details: | Mouse IgG2B Monoclonal Clone #OTI4G8 |
| Applications: | IHC, WB, Flow, ICC/IF, CyTOF-ready |
Loading...
| Reactivity: | Human, Mouse, Rat |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG2B Monoclonal Clone #666843 |
| Applications: | WB, Simple Western |
Loading...
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG2B Monoclonal Clone #666823 |
| Applications: | IHC |
Loading...
| Reactivity: | Human |
Loading...
| Reactivity: | Human, Mouse, Rat |
| Details: | Sheep IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Human, Mouse, Rat |
| Details: | Sheep IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Human, Mouse, Rat |
| Details: | Sheep IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Human, Mouse, Rat |
| Details: | Sheep IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Human, Mouse, Rat |
| Details: | Sheep IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Human, Mouse, Rat |
| Details: | Sheep IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Human, Mouse, Rat |
| Details: | Sheep IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Human, Mouse, Rat |
| Details: | Sheep IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG2B Monoclonal Clone #666843 |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG2B Monoclonal Clone #666843 |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG2B Monoclonal Clone #666843 |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG2B Monoclonal Clone #666843 |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG2B Monoclonal Clone #666843 |
| Applications: | WB |
Loading...



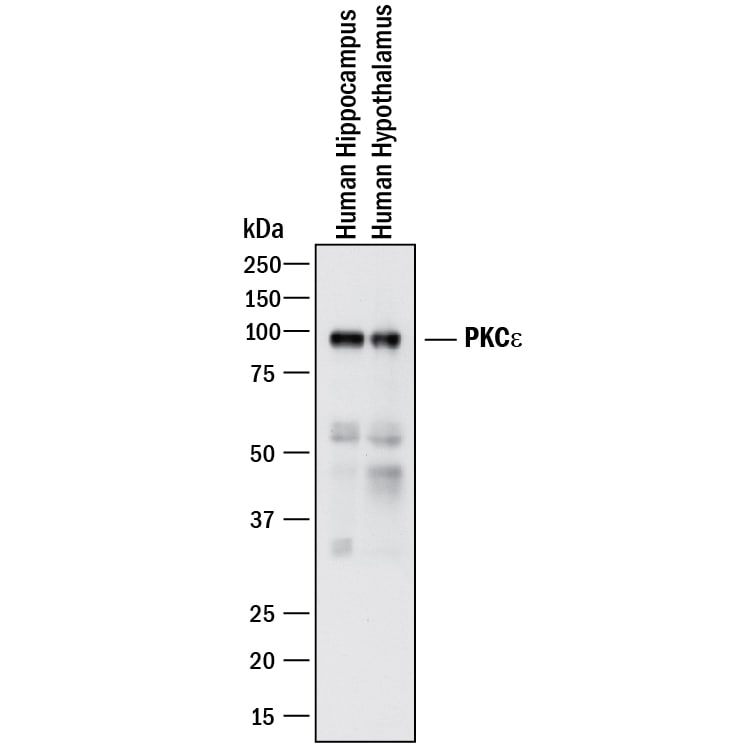

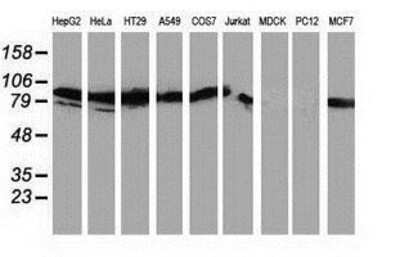
![Western Blot: PKC epsilon Antibody (OTI4G8)Azide and BSA Free [NBP2-73438] Western Blot: PKC epsilon Antibody (OTI4G8)Azide and BSA Free [NBP2-73438]](https://resources.rndsystems.com/images/products/PKC-epsilon-Antibody-OTI4G8-Azide-and-BSA-Free-Western-Blot-NBP2-73438-img0002.jpg)
![Western Blot: PKC epsilon [p Ser729] Antibody [NBP3-13312] Western Blot: PKC epsilon [p Ser729] Antibody [NBP3-13312]](https://resources.rndsystems.com/images/products/PKC-epsilon-[p-Ser729]-Antibody-Western-Blot-NBP3-13312-img0002.jpg)
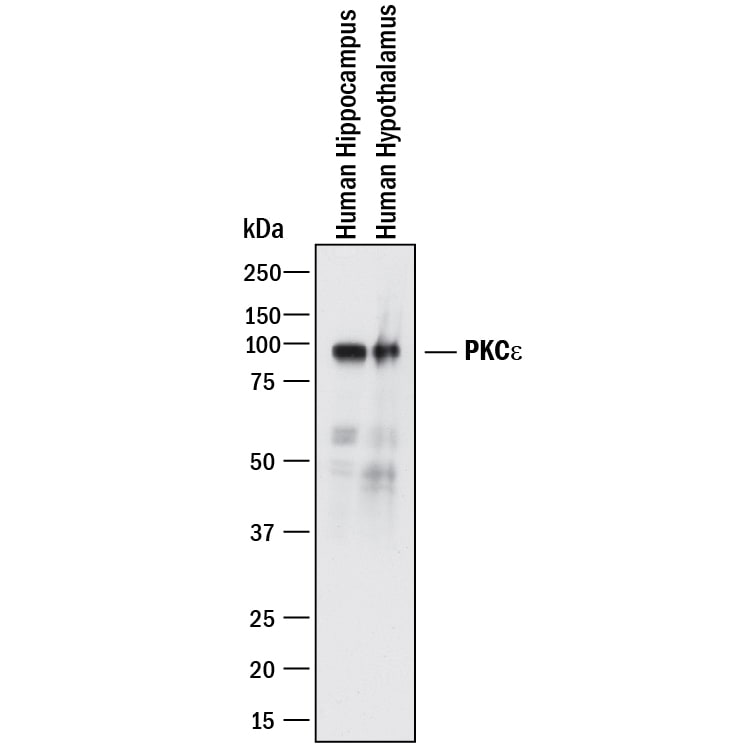
![Western Blot: PKC epsilon Antibody [NBP2-38531] Western Blot: PKC epsilon Antibody [NBP2-38531]](https://resources.rndsystems.com/images/products/PKC-epsilon-Antibody-Western-Blot-NBP2-38531-img0002.jpg)
![Western Blot: PKC epsilon Antibody [NBP2-19848] Western Blot: PKC epsilon Antibody [NBP2-19848]](https://resources.rndsystems.com/images/products/PKC-epsilon-Antibody-Western-Blot-NBP2-19848-img0001.jpg)
![Immunocytochemistry/ Immunofluorescence: PKC epsilon Antibody [NBP2-54970] Immunocytochemistry/ Immunofluorescence: PKC epsilon Antibody [NBP2-54970]](https://resources.rndsystems.com/images/products/PKC-epsilon-Antibody-Immunocytochemistry-Immunofluorescence-NBP2-54970-img0001.jpg)
![Immunocytochemistry/Immunofluorescence: PKC epsilon Antibody [NBP3-21224] - Immunocytochemistry/Immunofluorescence: PKC epsilon Antibody [NBP3-21224] -](https://resources.rndsystems.com/images/products/nbp3-21224_rabbit-pkc-epsilon-pab-4520231547285.jpg)