Having trouble trying to find the right product? Chat with our scientists below - we're here to help!
231 results for "S100A1" in 产品
S100A1 Products
S100A1 (also S100 alpha) is a 10 kDa member of the S100 family, EF-hand superfamily of Ca2+-binding proteins. It is expressed by striated muscle, endothelium and chondrocytes. Intracellularly, S100A1 regulates cytosolic calcium levels, and is part of the Hsp70/Hsp90 mutlichaperone complex that protects against protein denaturation. Extracellularly, S100A1 exists as both a homodimer and heterodimer with S100B, S100A4 and S100P, may bind to RAGE, and blocks apoptosis. Human S100A1 is 94 amino ...
| Reactivity: | Human, Mouse |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB |
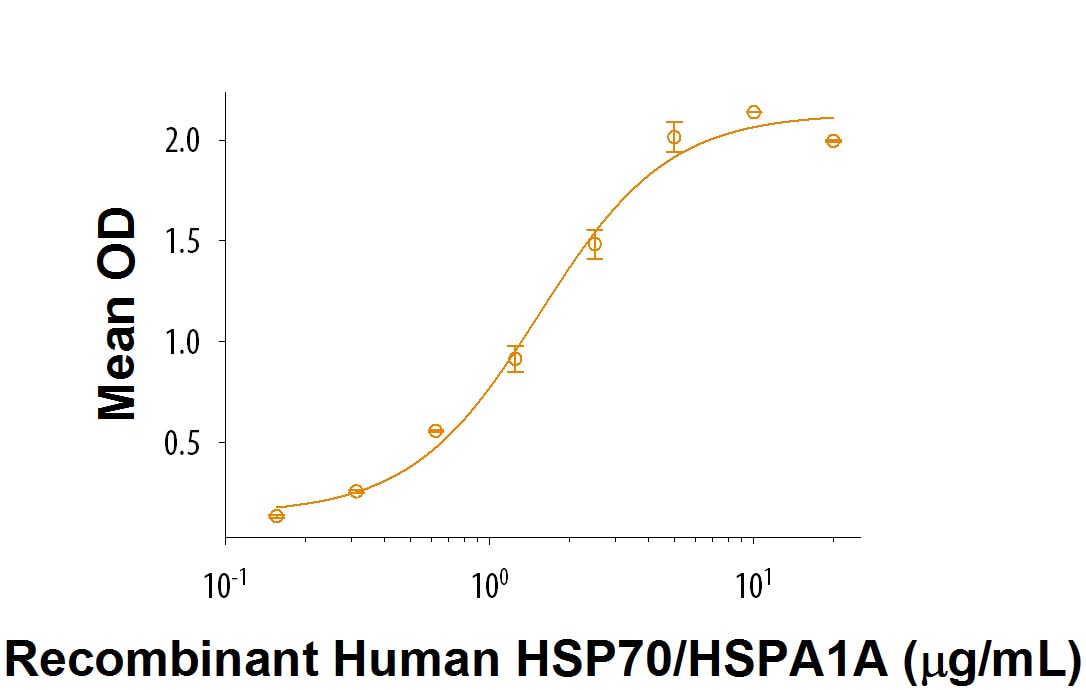
Loading...
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG1 kappa Monoclonal Clone #S100A1/1942 |
| Applications: | IHC, WB, Flow, ICC/IF, MA, + 1 More |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG1 kappa Monoclonal Clone #1D5 |
| Applications: | IHC, WB, ELISA |
Loading...
| Reactivity: | Human |
| Source: | E. coli |
| Accession #: | P23297 |
| Applications: | Bioactivity |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG1 kappa Monoclonal Clone #S100A1/1942 |
| Applications: | IHC, MA |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG1 kappa Monoclonal Clone #S100A1/1942 |
| Applications: | IHC, MA |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #2C8B8 |
| Applications: | IHC, WB, ELISA |
Loading...
| Reactivity: | Human, Mouse, Rat |
| Details: | Mouse IgG1 Monoclonal Clone #OTI6G4 |
| Applications: | WB, ELISA |
Loading...
| Reactivity: | Human, Mouse, Rat |
| Details: | Mouse IgG1 Monoclonal Clone #OTI6G4 |
| Applications: | WB, ELISA |
Loading...
Recombinant Monoclonal Antibody
| Reactivity: | Mouse |
| Details: | Rabbit IgG Monoclonal Clone #004 |
| Applications: | IHC |
Loading...
| Reactivity: | Human |
| Sensitivity: | 0.09 ng/mL |
| Assay Range: | 0.16 - 10 ng/mL |
| Applications: | ELISA |
Loading...
| Reactivity: | Mouse |
| Sensitivity: | 0.19 ng/mL |
| Assay Range: | 0.31 - 20 ng/mL |
| Applications: | ELISA |
Loading...
| Reactivity: | Rat |
| Sensitivity: | 18.75 pg/mL |
| Assay Range: | 31.25 - 2000 pg/mL |
| Applications: | ELISA |
Loading...
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Rabbit IgG Monoclonal Clone #D28-E |
| Applications: | IHC |
Loading...


![Western Blot: S100A1 Antibody [NBP1-87103] Western Blot: S100A1 Antibody [NBP1-87103]](https://resources.rndsystems.com/images/products/S100A1-Antibody-Western-Blot-NBP1-87103-img0016.jpg)


![Immunohistochemistry-Paraffin: S100A1 Antibody (S100A1/1942) - Azide and BSA Free [NBP2-47924] Immunohistochemistry-Paraffin: S100A1 Antibody (S100A1/1942) - Azide and BSA Free [NBP2-47924]](https://resources.rndsystems.com/images/products/S100A1-Antibody-S100A1-1012-Azide-and-BSA-Free-Immunohistochemistry-Paraffin-NBP2-47924-img0002.jpg)
![Western Blot: S100A1 Antibody (1D5) [H00006271-M01] Western Blot: S100A1 Antibody (1D5) [H00006271-M01]](https://resources.rndsystems.com/images/products/S100A1-Antibody-1D5-Western-Blot-H00006271-M01-img0004.jpg)
![Protein Array: S100A1 Antibody (S100A1/1942) [NBP2-59615] Protein Array: S100A1 Antibody (S100A1/1942) [NBP2-59615]](https://resources.rndsystems.com/images/products/S100A1-Antibody-S100A1-1942-Protein-Array-NBP2-59615-img0003.jpg)
![Immunohistochemistry-Paraffin: S100A1 Antibody (S100A1/1942) - Azide and BSA Free [NBP2-59617] Immunohistochemistry-Paraffin: S100A1 Antibody (S100A1/1942) - Azide and BSA Free [NBP2-59617]](https://resources.rndsystems.com/images/products/S100A1-Antibody-S100A1-1942-Azide-and-BSA-Free-Immunohistochemistry-Paraffin-NBP2-59617-img0003.jpg)
![Western Blot: S100A1 Antibody (2C8B8)BSA Free [NB110-60530] Western Blot: S100A1 Antibody (2C8B8)BSA Free [NB110-60530]](https://resources.rndsystems.com/images/products/S100A1-Antibody-2C8B8-Western-Blot-NB110-60530-img0004.jpg)
![Western Blot: S100A1 Antibody (OTI6G4) [NBP2-45658] Western Blot: S100A1 Antibody (OTI6G4) [NBP2-45658]](https://resources.rndsystems.com/images/products/S100A1-Antibody-6G4-Western-Blot-NBP2-45658-img0002.jpg)
![Western Blot: S100A1 Antibody (OTI6G4)Azide and BSA Free [NBP2-73967] Western Blot: S100A1 Antibody (OTI6G4)Azide and BSA Free [NBP2-73967]](https://resources.rndsystems.com/images/products/S100A1-Antibody-OTI6G4-Azide-and-BSA-Free-Western-Blot-NBP2-73967-img0001.jpg)
![Immunohistochemistry-Paraffin: S100A1 Antibody (004) [NBP2-90503] Immunohistochemistry-Paraffin: S100A1 Antibody (004) [NBP2-90503]](https://resources.rndsystems.com/images/products/S100A1-Antibody-004-Immunohistochemistry-Paraffin-NBP2-90503-img0002.jpg)
![ELISA: Human S100A1 ELISA Kit (Colorimetric) [NBP3-06949] - Human S100A1 ELISA Kit (Colorimetric)](https://resources.rndsystems.com/images/products/nbp3-06949_human-s100a1-elisa-kit-colorimetric-132202416133689.jpg)
![ELISA: Mouse S100A1 ELISA Kit (Colorimetric) [NBP3-06950] - Mouse S100A1 ELISA Kit (Colorimetric)](https://resources.rndsystems.com/images/products/nbp3-06950_mouse-s100a1-elisa-kit-colorimetric-132202416211362.jpg)
![ELISA: Rat S100A1 ELISA Kit (Colorimetric) [NBP3-06951] - Rat S100A1 ELISA Kit (Colorimetric)](https://resources.rndsystems.com/images/products/nbp3-06951_rat-s100a1-elisa-kit-colorimetric-132202416175610.jpg)
![Western Blot: S100A1 Overexpression Lysate [NBL1-15647] Western Blot: S100A1 Overexpression Lysate [NBL1-15647]](https://resources.rndsystems.com/images/products/S100A1-Overexpression-Lysate-Adult-Normal-Western-Blot-NBL1-15647-img0002.jpg)
![Immunohistochemistry-Paraffin: S100A1 Antibody (D28-E) [NBP1-30154] Immunohistochemistry-Paraffin: S100A1 Antibody (D28-E) [NBP1-30154]](https://resources.rndsystems.com/images/products/S100A-B-Antibody-D28-E-Immunohistochemistry-Paraffin-NBP1-30154-img0004.jpg)