169 results for "USP7" in 产品
USP7 Products
Ubiquitin Specific Peptidase 7 (USP7), also known as Herpes Virus-associated Ubiquitin Specific Protease (HAUSP), is a widely expressed deubiquitinating enzyme belonging to the peptidase C19 family. It has a predicted molecular weight of 130 kDa. Human USP7 is 1102 amino acids (aa) in length and shares 99% aa sequence identity with the mouse and rat orthologs. USP7 consists of a cysteine peptidase core (aa 208-560) that is flanked by an N-terminal TRAF-like domain (aa 50-205) and two C-terminal ...
| Reactivity: | Human, Mouse |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB, ICC/IF, IP |
| Reactivity: | Human |
| Source: | Sf 21 (baculovirus) |
| Accession #: | Q93009.2 |
| Applications: | Enzyme Activity |
| Reactivity: | Human, Mouse, Rat, Canine, Chicken, +2 More |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB |
| Reactivity: | Human, Mouse, Rat |
| Details: | Mouse IgG1 Monoclonal Clone #OTI1F12 |
| Applications: | IHC, WB, Flow |
| Reactivity: | Human, Mouse, Rat |
| Details: | Mouse IgG1 Monoclonal Clone #OTI1F12 |
| Applications: | IHC, WB, Flow, CyTOF-ready |
Recombinant Monoclonal Antibody
| Reactivity: | Human, Mouse, Rat |
| Details: | Rabbit IgG Monoclonal Clone #3J7G1 |
| Applications: | IHC, WB |
Recombinant Monoclonal Antibody
| Reactivity: | Human, Mouse |
| Details: | Rabbit IgG Monoclonal Clone #BLR072G |
| Applications: | IHC, WB, Flow, ICC/IF, IP |
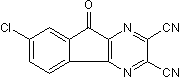
Selective USP7 inhibitor
| Chemical Name: | 7-Chloro-9-oxo-9H-indeno[1,2-b]pyrazine-2,3-dicarbonitrile |
| Purity: | ≥98% |
| Reactivity: | Human, Mouse, Rat, Canine, Chicken, +2 More |
| Details: | Rabbit IgG Polyclonal |
| Applications: | WB |
Recombinant Monoclonal Antibody
| Reactivity: | Human |
| Details: | Rabbit IgG Monoclonal Clone #7F10 |
| Applications: | IHC, WB, ELISA, ICC/IF |
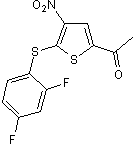
USP7 inhibitor
| Chemical Name: | 1-[5-[(2,4-Difluorophenyl)thio]-4-nitro-2-thienyl]-ethanone |
| Purity: | ≥98% |
Recombinant Monoclonal Antibody
| Reactivity: | Human, Mouse, Rat |
| Details: | Rabbit IgG Monoclonal Clone #S02-6B8 |
| Applications: | IHC, WB |
| Reactivity: | Human |
| Reactivity: | Human |
| Details: | Mouse IgG2B Monoclonal Clone #6512 |
| Applications: | WB, ICC/IF, IP |
Potent and selective USP7 Degrader (PROTAC®)
| Chemical Name: | 3-(1-(1-(2-(2,6-Dioxopiperidin-3-yl)-1,3-dioxoisoindolin-5-yl)piperidine-4-carbonyl)piperidin-4-yl)-N-(3-((4-hydroxy-1-((R)-3-phenylbutanoyl)piperidin-4-yl)methyl)-4-oxo-3,4-dihydroquinazolin-7-yl)propanamide |
| Purity: | ≥96% |
| Reactivity: | Human |
| Details: | Mouse IgG2A Monoclonal Clone #03 |
| Applications: | ELISA, Flow |
| Reactivity: | Human, Mouse, Rat |
| Details: | Rabbit IgG Monoclonal Clone #23GB2250 |
| Applications: | WB, ICC/IF, Mycoplasma |
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC |
| Reactivity: | Human |
| Applications: | WB |
| Reactivity: | Human |
| Details: | Mouse IgG2A Monoclonal Clone #06 |
| Applications: | IHC |
| Reactivity: | Human |
| Source: | E. coli |
| Applications: | AC |
| Reactivity: | Human, Mouse |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB, ICC/IF, IP |
| Reactivity: | Human, Mouse |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB, ICC/IF, IP |
| Reactivity: | Human, Mouse |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB, ICC/IF, IP |
| Reactivity: | Human, Mouse |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB, ICC/IF, IP |


![Western Blot: USP7 Antibody [NB100-513] Western Blot: USP7 Antibody [NB100-513]](https://resources.rndsystems.com/images/products/USP7-Antibody-Western-Blot-NB100-513-img0004.jpg)


![Western Blot: USP7 Antibody [NBP2-24641] Western Blot: USP7 Antibody [NBP2-24641]](https://resources.rndsystems.com/images/products/USP7-Antibody-Western-Blot-NBP2-24641-img0002.jpg)
![Western Blot: USP7 Antibody (OTI1F12) [NBP2-01544] Western Blot: USP7 Antibody (OTI1F12) [NBP2-01544]](https://resources.rndsystems.com/images/products/USP7-Antibody-1F12-Western-Blot-NBP2-01544-img0002.jpg)
![Western Blot: USP7 Antibody (OTI1F12)Azide and BSA Free [NBP2-74814] Western Blot: USP7 Antibody (OTI1F12)Azide and BSA Free [NBP2-74814]](https://resources.rndsystems.com/images/products/USP7-Antibody-OTI1F12-Azide-and-BSA-Free-Western-Blot-NBP2-74814-img0001.jpg)
![Western Blot: USP7 Antibody (3J7G1) [NBP3-16188] Western Blot: USP7 Antibody (3J7G1) [NBP3-16188]](https://resources.rndsystems.com/images/products/USP7-Antibody-3J7G1-Western-Blot-NBP3-16188-img0004.jpg)

![Western Blot: USP7 Antibody [NBP2-24642] Western Blot: USP7 Antibody [NBP2-24642]](https://resources.rndsystems.com/images/products/USP7-Antibody-Western-Blot-NBP2-24642-img0001.jpg)
![Immunohistochemistry: USP7 Antibody (7F10) [NBP3-26258] - USP7 Antibody (7F10)](https://resources.rndsystems.com/images/products/nbp3-26258_rabbit-usp7-mab-7f10-262202415253324.jpg)
![Western Blot: USP7 Antibody (S02-6B8) [NBP3-19800] Western Blot: USP7 Antibody (S02-6B8) [NBP3-19800]](https://resources.rndsystems.com/images/products/USP7-Antibody-S02-6B8-Western-Blot-NBP3-19800-img0002.jpg)
![Western Blot: USP7 Antibody (6512) [NBP2-43619] Western Blot: USP7 Antibody (6512) [NBP2-43619]](https://resources.rndsystems.com/images/products/USP7-Antibody-6512-Western-Blot-NBP2-43619-img0001.jpg)

![Flow Cytometry: USP7 Antibody (03) [NBP3-06427] Flow Cytometry: USP7 Antibody (03) [NBP3-06427]](https://resources.rndsystems.com/images/products/USP7-Antibody-03-Flow-Cytometry-NBP3-06427-img0002.jpg)
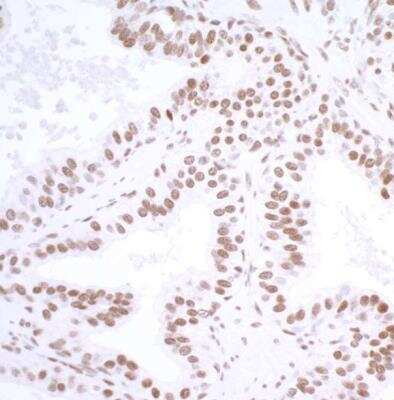
![Immunohistochemistry-Paraffin: USP7 Antibody [NBP1-83293] Immunohistochemistry-Paraffin: USP7 Antibody [NBP1-83293]](https://resources.rndsystems.com/images/products/USP7-Antibody-Immunohistochemistry-Paraffin-NBP1-83293-img0004.jpg)
![Western Blot: USP7 Overexpression Lysate [NBL1-17671] Western Blot: USP7 Overexpression Lysate [NBL1-17671]](https://resources.rndsystems.com/images/products/USP7-Overexpression-Lysate-Adult-Normal-Western-Blot-NBL1-17671-img0002.jpg)
![Immunohistochemistry-Paraffin: USP7 Antibody (06) [NBP3-06428] Immunohistochemistry-Paraffin: USP7 Antibody (06) [NBP3-06428]](https://resources.rndsystems.com/images/products/USP7-Antibody-06-Immunohistochemistry-Paraffin-NBP3-06428-img0001.jpg)
![Product Feature: CoraFluor Probes for TR-FRET USP7 Antibody [CoraFluor™ 1]](https://resources.rndsystems.com/images/products/nb100-513cl1_rabbit-usp7-pab-corafluor-1-99202510284221.png)