AP 18
Tocris Bioscience | Catalog # 3296

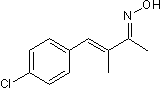
Product Description
AP 18 is a reversible TRPA1 channel blocker (IC50 values are 3.1 and 4.5 μM at human and mouse TRPA1 respectively). Blocks cinnamaldehyde-induced but not capsaicin-induced nociception and reverses mechanical hyperalgesia in vivo. Also blocks TRPA1 pore dilation (IC50 = 10.3 μM for the inhibition of Yo-Pro uptake).
Product Specifications for AP 18
Molecular Weight
Formula
Storage
Purity
Chemical Name
CAS Number
PubChem ID
InChI Key
SMILES
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Solubility
| Solvent | Max Conc. mg/mL | Max Conc. mM | |
|---|---|---|---|
| Solubility | |||
| DMSO | 20.97 | 100 | |
| Ethanol | 20.97 | 100 |
Preparing Stock Solutions for AP 18
The following data is based on the product molecular weight 209.67.
Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which all affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 1 mM | 4.77 mL | 23.85 mL | 47.69 mL |
| 5 mM | 0.95 mL | 4.77 mL | 9.54 mL |
| 10 mM | 0.48 mL | 2.38 mL | 4.77 mL |
| 50 mM | 0.10 mL | 0.48 mL | 0.95 mL |
Calculators
Background References
References are publications that support the biological activity of the product. See our Citations tab to view 17 publications citing the usage of this product.
- Chen Pore dilation occurs in TRPA1 but not in TRPM8 channels. Mol.Pain 2009 PMID: 19159452
- Taylor-Clark Nitrooleic acid, an endogenous product of nitrative stress, activates nociceptive sensory nerves via the direct activation of TRPA1. Mol.Pharmacol. 2009 PMID: 19171673
- Petrus A role of TRPA1 in mechanical hyperalgesia is revealed by pharmacological inhibition. Mol.Pain 2007 PMID: 18086313
Product Documents for AP 18
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for AP 18
For research use only
Related Research Areas
Citations for AP 18
Customer Reviews for AP 18
There are currently no reviews for this product. Be the first to review AP 18 and earn rewards!
Have you used AP 18?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
