BMS 299897
Tocris Bioscience | Catalog # 2870

Product Description
BMS 299897 is an orally active, potent γ-secretase inhibitor (IC50 = 12 nM). Inhibits Aβ40 and Aβ42 formation in vitro (IC50 values are 7.4 and 7.9 nM respectively) and reduces Aβ in the brain, plasma and cerebrospinal fluid in vivo. Exhibits no Notch toxicity. Brain penetrant.
Product Specifications for BMS 299897
Molecular Weight
Formula
Storage
Purity
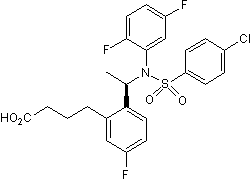
Chemical Name
CAS Number
PubChem ID
InChI Key
SMILES
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Solubility
| Solvent | Max Conc. mg/mL | Max Conc. mM | |
|---|---|---|---|
| Solubility | |||
| DMSO | 51.19 | 100 | |
| Ethanol | 51.19 | 100 |
Preparing Stock Solutions for BMS 299897
The following data is based on the product molecular weight 511.94.
Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which all affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 1 mM | 1.95 mL | 9.77 mL | 19.53 mL |
| 5 mM | 0.39 mL | 1.95 mL | 3.91 mL |
| 10 mM | 0.20 mL | 0.98 mL | 1.95 mL |
| 50 mM | 0.04 mL | 0.20 mL | 0.39 mL |
Calculators
Background References
References are publications that support the biological activity of the product. See our Citations tab to view 8 publications citing the usage of this product.
- Anderson Reductions in β-amyloid concentrations in vivo by the γ-secretase inhibitors BMS-289948 and BMS-299897. Biochem.Pharmacol. 2005 PMID: 15670587
- Goldstein Ex vivo occupancy of the γ-secretase inhibitors correlates with brain β-amyloid peptide reduction in Tg2576 mice. J.Pharmacol.Exp.Ther. 2007 PMID: 17640949
- Barten Dynamics of β-Amyloid reductions in brain, cerebrospinal fluid, and plasma of β-amyloid precursor protein transgenic mice treated with a γ-secretase inhibitor. J.Pharmacol.Exp.Ther. 2005 PMID: 15452193
Product Documents for BMS 299897
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for BMS 299897
For research use only
Citations for BMS 299897
Customer Reviews for BMS 299897 (2)
Have you used BMS 299897?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
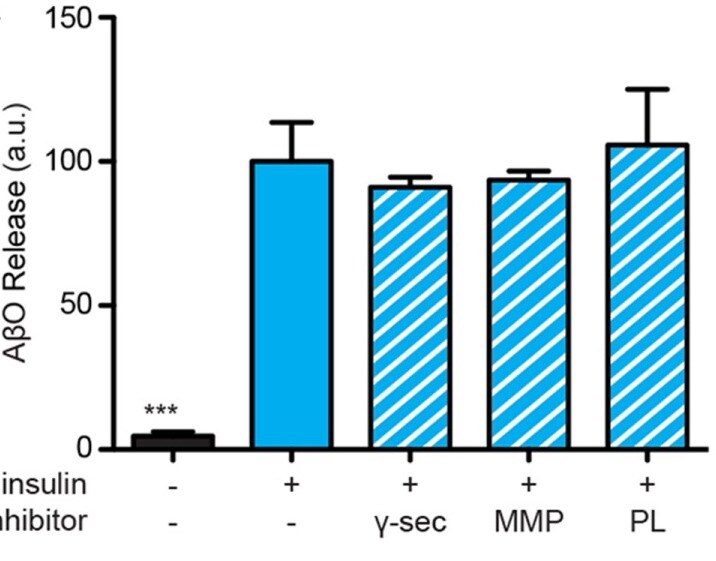
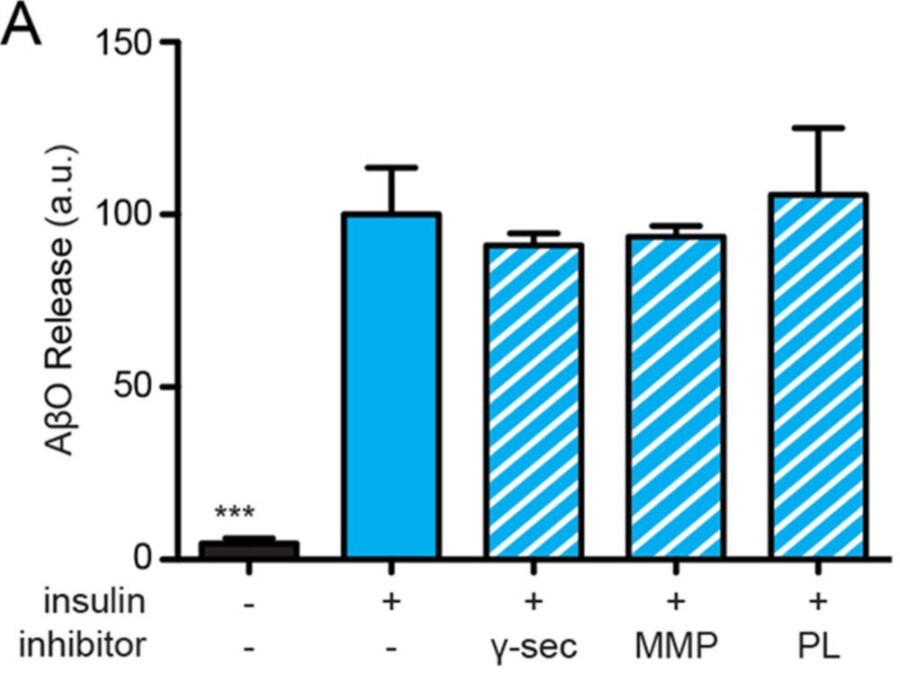
Species: RatAssay Type: In VitroCell Line/Tissue: Hippocampal neuronVerified Customer | Posted 03/26/2020gamma -secretase (100 nM BMS 299897 and 250 nM DAPT)
-
Species: HumanAssay Type: In VitroCell Line/Tissue: Hippocampal neuronVerified Customer | Posted 10/13/2019Using more specific inhibitors, we ruled out the involvement of gamma -secretase (100 nM BMS 299897 and 250 nM DAPT) or matrix metalloproteinases 1, 2, 3, 7, 9, 14, and 17 (0.1–1 μM batimastat and marimastat), as their inhibitors failed to alter A beta O release
There are no reviews that match your criteria.