Carfilzomib
Tocris Bioscience | Catalog # 7188

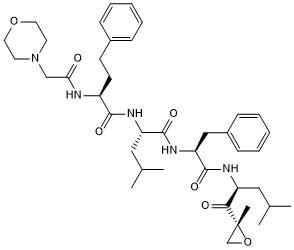
Key Product Details
Description
Alternative Names
Product Description
Carfilzomib is a potent irreversible proteasome inhibitor. Preferentially inhibits the chymotrypsin-like β5 subunit of the constitutive 20S proteasome (IC50 = 5.2 nM) and the β5i subunit of the immunoproteasome 20Si (LMP7; IC50 = 14 nM) in vitro with minimal cross-reactivity to other proteases. Exhibits little or no effect on PGPH and T-L activities. Activates prosurvival autophagy and induces cell apoptosis. Acts synergistically with dexamethasone (Cat. No. 1126). Suppresses tumor growth in an in vivo xenograft model. Decreases bone resorption and enhances bone formation in non-tumor bearing mice.
Product Specifications for Carfilzomib
Molecular Weight
Formula
Storage
Purity
Chemical Name
CAS Number
PubChem ID
InChI Key
SMILES
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Solubility
| Solvent | Max Conc. mg/mL | Max Conc. mM | |
|---|---|---|---|
| Solubility | |||
| DMSO | 71.99 | 100 | |
| Ethanol | 14.4 | 20 |
Preparing Stock Solutions for Carfilzomib
The following data is based on the product molecular weight 719.92.
Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which all affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 1 mM | 1.39 mL | 6.95 mL | 13.89 mL |
| 5 mM | 0.28 mL | 1.39 mL | 2.78 mL |
| 10 mM | 0.14 mL | 0.69 mL | 1.39 mL |
| 50 mM | 0.03 mL | 0.14 mL | 0.28 mL |
Calculators
Background References
References are publications that support the biological activity of the product. See our Citations tab to view 5 publications citing the usage of this product.
- Wang Fast identification of possible drug treatment of coronavirus disease-19 (COVID-19) through computational drug repurposing study. J.Chem.Inf.Model 2020 PMID: 32315171
- Zhou Design and synthesis of an orally bioavailable and selective peptide epoxyketone proteasome inhibitor (PR-047). J.Med.Chem. 2009 PMID: 19348473
- Dou Overview of proteasome inhibitor-based anti-cancer therapies: perspective on bortezomib and second generation proteasome inhibitors versus future generation inhibitors of ubiquitin-proteasome system. Curr.Cancer Drug Targets 2014 PMID: 25092212
- Demo Antitumor activity of PR-171, a novel irreversible inhibitor of the proteasome. Cancer Res. 2007 PMID: 17616698
Product Documents for Carfilzomib
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for Carfilzomib
For research use only
Related Research Areas
Citations for Carfilzomib
Customer Reviews for Carfilzomib (1)
Have you used Carfilzomib?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
-
Species: HumanAssay Type: In VitroVerified Customer | Posted 10/13/2022Combinatorial treatments of carfilzomib with chemotherapeutics led to synergistic apoptotic phenotype in vitro.
There are no reviews that match your criteria.
