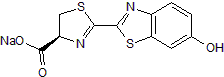
D-Luciferin sodium salt
Tocris Bioscience | Catalog # 5427

Key Product Details
Description
Product Description
D-Luciferin sodium salt is a firefly luciferase substrate (Km = ~2 μM). Used in many standard bioluminescence imaging (BLI) techniques, including monitoring tumour growth, and investigating intracellular signaling activity in vitro and in vivo. Used with MC3 LNP Luciferase (Cat. No. 8889) for in vitro and in vivo bioluminescence LNP detection.Cell permeable. Emission maximum = 562 nm.Optical Data

| Emission Color | Green |
| λem | 562 nm |
| Cell Permeable | Yes |
| Application | Bioluminescence Imaging (BLI) Techniques, including monitoring tumor growth |
Spectra Viewer
Plan Your Experiments
Use our spectra viewer to interactively plan your experiments, assessing multiplexing options. View the excitation and emission spectra for our fluorescent dye range and other commonly used dyes.
Spectra Viewer
Product Specifications for D-Luciferin sodium salt
Molecular Weight
Formula
Storage
Purity
Chemical Name
CAS Number
PubChem ID
InChI Key
SMILES
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Solubility
| Solvent | Max Conc. mg/mL | Max Conc. mM | |
|---|---|---|---|
| Solubility | |||
| water | 30.23 | 100 | |
| DMSO | 30.23 | 100 |
Preparing Stock Solutions for D-Luciferin sodium salt
The following data is based on the product molecular weight 302.30.
Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which all affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 1 mM | 3.31 mL | 16.54 mL | 33.08 mL |
| 5 mM | 0.66 mL | 3.31 mL | 6.62 mL |
| 10 mM | 0.33 mL | 1.65 mL | 3.31 mL |
| 50 mM | 0.07 mL | 0.33 mL | 0.66 mL |
Calculators
Background References
References are publications that support the biological activity of the product. See our Citations tab to view 9 publications citing the usage of this product.
- Keyaerts Dynamic bioluminescence imaging for quantitative tumour burden assessment using IV or IP administration of D: -luciferin: effect on intensity, time kinetics and repeatability of photon emission. Eur.J.Nucl.Med.Mol.Imaging. 2008 PMID: 18180921
Product Documents for D-Luciferin sodium salt
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for D-Luciferin sodium salt
For research use only
Citations for D-Luciferin sodium salt
Customer Reviews for D-Luciferin sodium salt (1)
Have you used D-Luciferin sodium salt?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
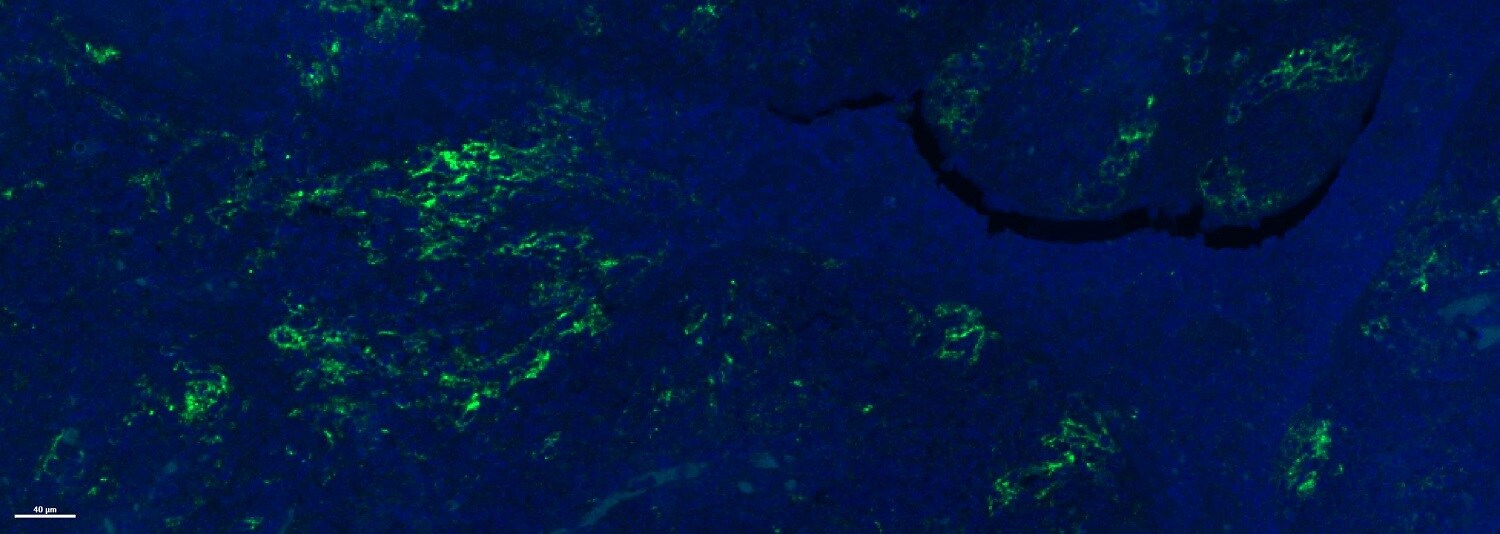
Species: HumanVerified Customer | Posted 08/18/2019D-Luciferin was used to assess ATP availability in cellular assays.The fluorescent images were distinguishable and publish worthy.
There are no reviews that match your criteria.