Iloprost
Tocris Bioscience | Catalog # 2038

Key Product Details
Description
Alternative Names
Product Description
Iloprost is a prostacyclin (PGI2) analog that binds with high affinity to IP, EP1 and EP3 receptors (Ki values are 11, 11, 56, 284, 619, 1035, 1870 and 6487 nM for IP, EP1, EP3, EP4, FP, DP, EP2 and TP receptors respectively). Inhibits platelet aggregation induced by collagen, thrombin and ADP (IC50 values are 0.24, 0.71 and 1.07 nM respectively).
Product Specifications for Iloprost
Molecular Weight
Formula
Storage
Purity
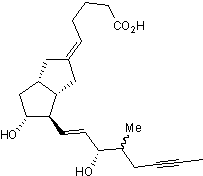
Chemical Name
CAS Number
PubChem ID
InChI Key
SMILES
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Solubility
| Solubility | Soluble in methyl acetate (supplied pre-dissolved - 5mg/ml) |
Calculators
Background References
References are publications that support the biological activity of the product. See our Citations tab to view 21 publications citing the usage of this product.
- Abramovitz The utilization of recombinant prostanoid receptors to determine the affinities and selectivities of prostaglandins and related analogs. Biochim.Biophys.Acta 2000 PMID: 10634944
- Schror The antiplatelet and cardiovascular actions of a new carbacyclin derivative (ZK 36 374) - equipotent to PGI2 in vitro. Naunyn-Schmied.Arch.Pharmacol.
- Della Bella Novel mode of action of iloprost: in vitro down-regulaton of endothelial cell adhesion molecules. Prostaglandins Other Lipid Mediat. 2001 PMID: 11403500
Product Documents for Iloprost
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for Iloprost
For research use only
Related Research Areas
Citations for Iloprost
Customer Reviews for Iloprost
There are currently no reviews for this product. Be the first to review Iloprost and earn rewards!
Have you used Iloprost?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
