L 006235
Tocris Bioscience | Catalog # 3066

Key Product Details
Description
Product Description
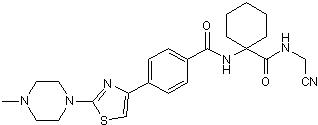
L 006235 is a potent, reversible cathepsin K inhibitor (IC50 = 0.25 nM) that displays > 4000-fold selectivity over cathepsins B, L and S. Displays reduced selectivity in cell-based assays possibly due to lysosomal accumulation. Reduces collagen breakdown and promotes bone deposition in vivo. Orally active and has intrinsic fluorescence.
Product Specifications for L 006235
Molecular Weight
Formula
Storage
Purity
Chemical Name
CAS Number
PubChem ID
InChI Key
SMILES
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Solubility
| Solvent | Max Conc. mg/mL | Max Conc. mM | |
|---|---|---|---|
| Solubility | |||
| DMSO | 46.66 | 100 |
Preparing Stock Solutions for L 006235
The following data is based on the product molecular weight 466.60.
Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which all affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 1 mM | 2.14 mL | 10.72 mL | 21.43 mL |
| 5 mM | 0.43 mL | 2.14 mL | 4.29 mL |
| 10 mM | 0.21 mL | 1.07 mL | 2.14 mL |
| 50 mM | 0.04 mL | 0.21 mL | 0.43 mL |
Calculators
Background References
References are publications that support the biological activity of the product. See our Citations tab to view 3 publications citing the usage of this product.
- Falgueyret Lysosomotropism of basic cathepsin K inhibitors contributes to increased cellular potencies against off-target cathepsins and reduced functional selectivity. J.Med.Chem. 2005 PMID: 16302795
- Desmarais Effects of cathepsin K inhibitors basicity on in vivo off-target activities. Mol.Pharmacol. 2008 PMID: 17940194
- Palmer Design and synthesis of tri-ring P3 benamide-containing aminonitriles as potent, selective, orally effective inhibitors of cathepsin K. J.Med.Chem. 2005 PMID: 16302794
Product Documents for L 006235
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for L 006235
For research use only
Related Research Areas
Citations for L 006235
Customer Reviews for L 006235
There are currently no reviews for this product. Be the first to review L 006235 and earn rewards!
Have you used L 006235?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
