ML 193
Tocris Bioscience | Catalog # 4860

Key Product Details
Description
Alternative Names
Product Description
ML 193 is a selective GPR55 antagonist (IC50 = 221 nM). Exhibits >27-, >145- and >145-fold selectivity for GPR55 over CB1, GPR35 and CB2, respectively. Inhibits ERK signaling in vitro.
Product Specifications for ML 193
Molecular Weight
Formula
Storage
Purity
Chemical Name
CAS Number
PubChem ID
InChI Key
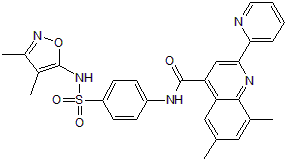
SMILES
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Solubility
| Solvent | Max Conc. mg/mL | Max Conc. mM | |
|---|---|---|---|
| Solubility | |||
| DMSO | 52.76 | 100 |
Preparing Stock Solutions for ML 193
The following data is based on the product molecular weight 527.59.
Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which all affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 1 mM | 1.90 mL | 9.48 mL | 18.95 mL |
| 5 mM | 0.38 mL | 1.90 mL | 3.79 mL |
| 10 mM | 0.19 mL | 0.95 mL | 1.90 mL |
| 50 mM | 0.04 mL | 0.19 mL | 0.38 mL |
Calculators
Background References
References are publications that support the biological activity of the product. See our Citations tab to view 10 publications citing the usage of this product.
- Kotsikorou Identification of the GPR55 antagonist binding site using a novel set of high-potency GPR55 selective ligands. Biochemistry 2013 PMID: 24274581
- Heynen-Genel Screening for selective ligands for GPR55 - antagonists. Probe Reports from the NIH Molecular Libraries Program 2010 PMID: 22091481
Product Documents for ML 193
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for ML 193
For research use only
Citations for ML 193
Customer Reviews for ML 193 (1)
Have you used ML 193?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
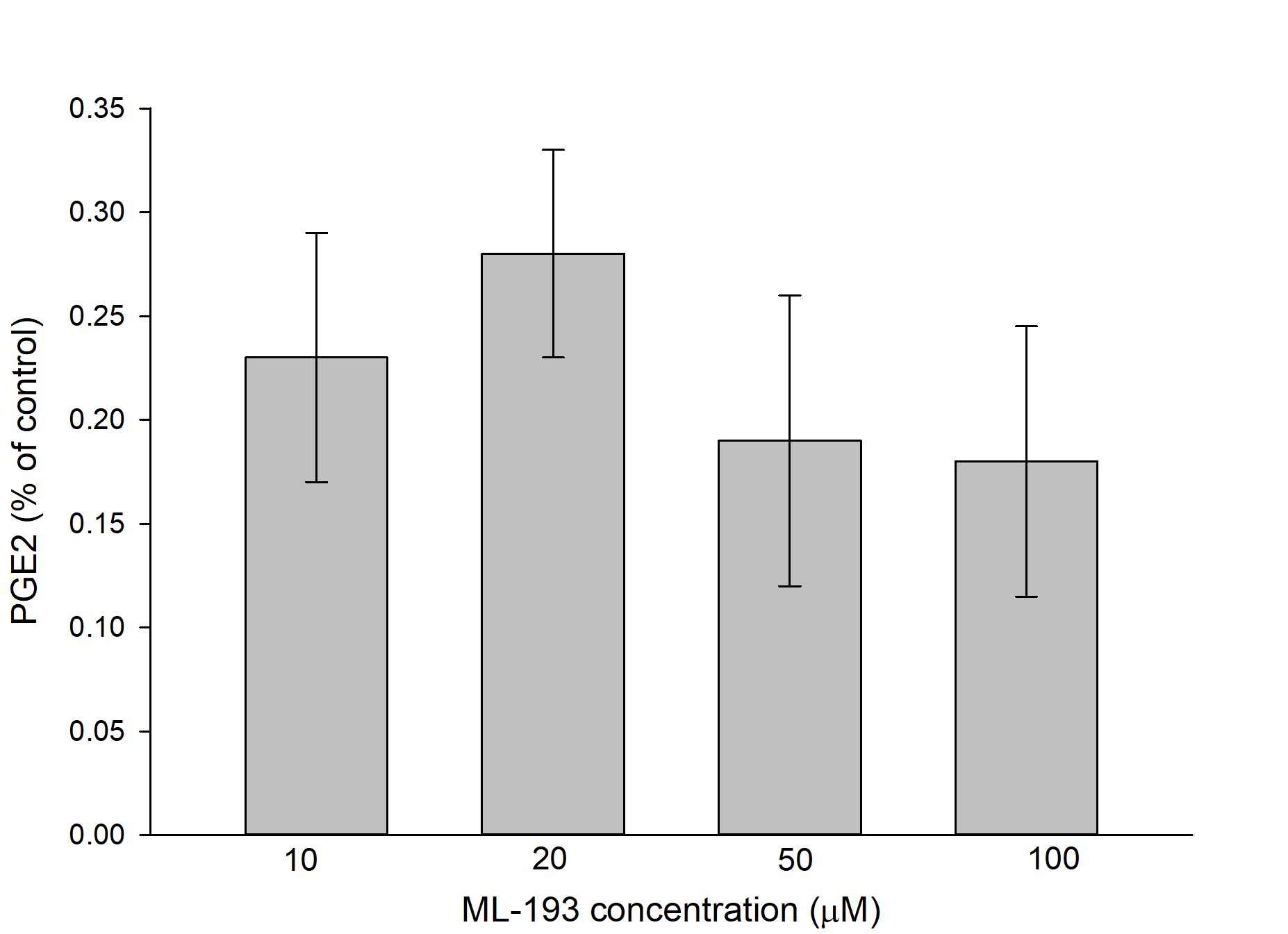
Species: MouseAssay Type: In VitroCell Line/Tissue: HippocampusVerified Customer | Posted 09/21/2019Soluble in DMSO, perfectly mixes with artificial cerebro-spinal fluid.ML-193 was used to study the effect of receptor GPR-55 on release of prostaglandin E2 from hippocampal microglia. ML-193 triggered highly significant lowering of the prostaglandin release compared to control; however, difference between the effects of four tested doses was non-significant (see figure).
There are no reviews that match your criteria.