Mouse CCL2 is a member of the beta (C-C) subfamily of chemokines. The mouse CCL2 gene was initially identified as a platelet-derived growth factor-inducible gene in mouse fibroblasts. Mouse CCL2 cDNA encodes a 148 amino acid (aa) residue with a putative 23 aa signal peptide that is cleaved to generate the mature protein. Mouse CCL2 shares 82% amino acid sequence identity with rat CCL2. Mouse CCL2 also shares 55% amino acid sequence identity with human MCP-1. Compared to human MCP-1, mouse CCL2 has a 49 aa residue extension at the carboxy-terminus. When a DNA sequence encoding the 125 aa residue of the mature CCL2 protein was expressed in E. coli at R&D Systems, the purified protein had the predicted N-terminus but a mass of 8525 Da. The truncation of most of the C-terminal extension could be due either to purification artifact or to post-translational modification. The truncated recombinant CCL2 has a potency similar to that of human MCP-1 in the monocyte chemotaxis assay. Mouse CCL2 has full activity on human cells while human MCP-1 has limited activity on mouse cells.
Mouse CCL2/JE/MCP‑1 Antibody
R&D Systems | Catalog # MAB479R
Recombinant Monoclonal Antibody.

Discontinued Product
MAB479R has been discontinued.
View all CCL2/JE/MCP-1 products.
Key Product Details
Species Reactivity
Mouse
Applications
Flow Cytometry
Label
Unconjugated
Antibody Source
Recombinant Monoclonal Rat IgG2B Clone # 123616R
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant mouse CCL2
Gln24-Arg96
Accession # P10148
Gln24-Arg96
Accession # P10148
Specificity
Detects mouse CCL2 in direct ELISAs.
Clonality
Monoclonal
Host
Rat
Isotype
IgG2B
Scientific Data Images for Mouse CCL2/JE/MCP‑1 Antibody
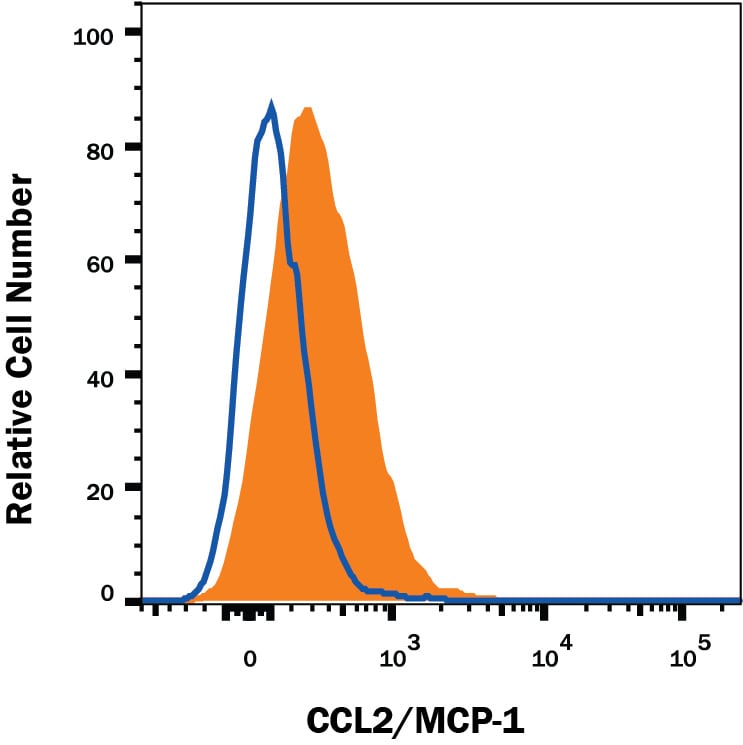
Detection of CCL2/JE/MCP‑1 in Mouse Splenocytes by Flow Cytometry.
Mouse splenocytes either unstimulated (open histogram) or stimulated with 1 ug/mL LPS overnight and 3 uM monensin for 2 hours (filled histogram) were stained with Recombinant Rat Anti-Mouse CCL2/JE/MCP-1 Monoclonal Antibody (Catalog # MAB479R), followed by Phycoerythrin-conjugated Anti-Rat IgG Secondary Antibody (Catalog # F0105B). To facilitate intracellular staining, cells were fixed with Flow Cytometry Fixation Buffer (Catalog # FC004) and permeabilized with Flow Cytometry Permeabilization/Wash Buffer I (Catalog # FC005). View our protocol for Staining Intracellular Molecules.Applications for Mouse CCL2/JE/MCP‑1 Antibody
Application
Recommended Usage
Flow Cytometry
0.25 µg/106 cells
Sample: Mouse splenocytes treated with LPS and monensin
Sample: Mouse splenocytes treated with LPS and monensin
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Protein A or G purified from cell culture supernatant
Reconstitution
Reconstitute at 0.5
mg/mL in sterile PBS.
For liquid material, refer to CoA for concentration.
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: CCL2/JE/MCP-1
References
- Rollins, B.J. et al. (1988) Proc. Natl. Acad. Sci. USA 85:3738.
- Gu, L. et al. (1999) Chem. Immunol. 72:7.
- Luini, W. et al. (1994) Cytokine 6:28.
Alternate Names
GDCF-2, HC11, HSMCR30, MCAF, MCP-1, SMC-CF
Gene Symbol
CCL2
UniProt
Additional CCL2/JE/MCP-1 Products
Product Documents for Mouse CCL2/JE/MCP‑1 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse CCL2/JE/MCP‑1 Antibody
For research use only
Customer Reviews for Mouse CCL2/JE/MCP‑1 Antibody
There are currently no reviews for this product. Be the first to review Mouse CCL2/JE/MCP‑1 Antibody and earn rewards!
Have you used Mouse CCL2/JE/MCP‑1 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- View all Protocols, Troubleshooting, Illustrated assays and Webinars




