SPARC, (Secreted Protein, Acidic and Rich in Cysteine) also known as Osteonectin or BM-40. It is the founding member of a family of secreted matricellular proteins with similar domain structure. The 295 amino acid (aa), 43-45 kDa protein contains a 46 aa N-terminal acidic region that binds calcium, a 23 aa follistatin-like domain containing Kazal-like sequences, and a 148 aa C-terminal extracellular calcium (EC) binding domain with two EF-hand motifs (1‑5). Crystal structure shows that residues implicated in cell binding, inhibition of cell spreading, and disassembly of focal adhesions, cluster on one face of SPARC, while a collagen binding epitope, and an N-glycosylation site are opposite this face (6). SPARC is produced by fibroblasts, capillary endothelial cells, platelets and macrophages, especially in areas of tissue morphogenesis and remodeling (3, 7). It is also secreted by astrocytes where it participates in synapse formation (8). SPARC shows context-specific effects, but generally inhibits adhesion, membrane spreading and cell proliferation, while promoting collagen matrix formation (3‑5). For endothelial cells, SPARC disrupts focal adhesions and binds and sequesters PDGF and VEGF (3‑5). SPARC is abundantly expressed in bone, where it promotes osteoblast differentiation and inhibits adipogenesis (5, 9). SPARC is potentially cleaved by metalloproteinases, producing an angiogenic peptide that includes the copper-binding sequence KGHK (7). Paradoxically, SPARC is highly expressed in many tumor types, yet expression mainly decreases the likelihood of metastasis and confers sensitivity to chemotherapy and radiation (4, 10, 11). Stabilin-1, which is expressed on alternately activated macrophages, is the first SPARC receptor to be identified. It binds the SPARC EC domain and mediates endocytosis for degradation (12). Mature mouse SPARC shows 97%, 92%, 92%, 92% and 83% aa identity with rat, human, dog, cow and chick SPARC, respectively.
Mouse SPARC Alexa Fluor® 488‑conjugated Antibody
R&D Systems | Catalog # IC942G

Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Ala18-Ile302
Accession # P07214
Specificity
Clonality
Host
Isotype
Scientific Data Images for Mouse SPARC Alexa Fluor® 488‑conjugated Antibody
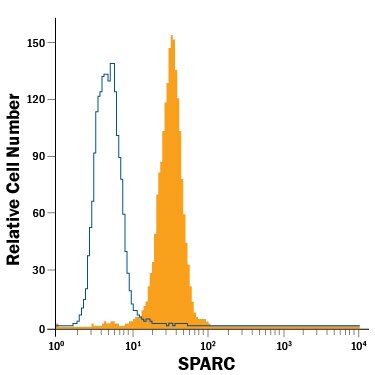
Detection of SPARC in Balb/3T3 Mouse Cell Line by Flow Cytometry.
Balb/3T3 mouse embryonic fibroblast cell line was stained with Rat Anti-Mouse SPARC Alexa Fluor® 488-conjugated Monoclonal Antibody (Catalog # IC942G, filled histogram) or isotype control antibody (Catalog # IC013G, open histogram). To facilitate intracellular staining, cells were fixed with Flow Cytometry Fixation Buffer (Catalog # FC004) and permeabilized with Flow Cytometry Permeabilization/Wash Buffer I (Catalog # FC005). View our protocol for Staining Intracellular Molecules.Applications for Mouse SPARC Alexa Fluor® 488‑conjugated Antibody
Intracellular Staining by Flow Cytometry
Sample: Balb/3T3 mouse embryonic fibroblast cell line fixed with Flow Cytometry Fixation Buffer (Catalog # FC004) and permeabilized with Flow Cytometry Permeabilization/Wash Buffer I (Catalog # FC005)
Spectra Viewer
Plan Your Experiments
Use our spectra viewer to interactively plan your experiments, assessing multiplexing options. View the excitation and emission spectra for our fluorescent dye range and other commonly used dyes.
Spectra ViewerFlow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Formulation
Shipping
Stability & Storage
Background: SPARC
References
- Lankat-Buttgereit, B. et al. (1988) FEBS Lett. 236:352.
- Bradshaw, A.D. (2012) Intl. J. Biochem. Cell Biol. 44:480.
- Sage, H. et al. (1989) J. Cell Biol. 109:341.
- Framson, P.E. and E.H. Sage (2004) J. Cell. Biochem. 92:679.
- Alford, A.I. and K.D. Hankenson (2006) Bone 38:749.
- Hohenester, E. et al. (1997) EMBO J. 16:3778.
- Sage, E.H. et al. (2003) J. Biol. Chem. 278:37849.
- Jones, E.V. et al. (2008) J. Neurosci. 31:4154.
- Delany, A.M. et al. (2003) Endocrinology 144:2588.
- Koblinski, J.E. et al. (2005) Cancer Res. 65:7370.
- Tai, I.T. et al. (2005) J. Clin. Invest. 115:1492.
- Kzhyshkowska, J. et al. (2006) J. Immunol. 176:5825.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional SPARC Products
Product Documents for Mouse SPARC Alexa Fluor® 488‑conjugated Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse SPARC Alexa Fluor® 488‑conjugated Antibody
This product is provided under an agreement between Life Technologies Corporation and R&D Systems, Inc, and the manufacture, use, sale or import of this product is subject to one or more US patents and corresponding non-US equivalents, owned by Life Technologies Corporation and its affiliates. The purchase of this product conveys to the buyer the non-transferable right to use the purchased amount of the product and components of the product only in research conducted by the buyer (whether the buyer is an academic or for-profit entity). The sale of this product is expressly conditioned on the buyer not using the product or its components (1) in manufacturing; (2) to provide a service, information, or data to an unaffiliated third party for payment; (3) for therapeutic, diagnostic or prophylactic purposes; (4) to resell, sell, or otherwise transfer this product or its components to any third party, or for any other commercial purpose. Life Technologies Corporation will not assert a claim against the buyer of the infringement of the above patents based on the manufacture, use or sale of a commercial product developed in research by the buyer in which this product or its components was employed, provided that neither this product nor any of its components was used in the manufacture of such product. For information on purchasing a license to this product for purposes other than research, contact Life Technologies Corporation, Cell Analysis Business Unit, Business Development, 29851 Willow Creek Road, Eugene, OR 97402, Tel: (541) 465-8300. Fax: (541) 335-0354.
For research use only
Citations for Mouse SPARC Alexa Fluor® 488‑conjugated Antibody
Customer Reviews for Mouse SPARC Alexa Fluor® 488‑conjugated Antibody
There are currently no reviews for this product. Be the first to review Mouse SPARC Alexa Fluor® 488‑conjugated Antibody and earn rewards!
Have you used Mouse SPARC Alexa Fluor® 488‑conjugated Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Associated Pathways