PFI 3
Tocris Bioscience | Catalog # 5072

Key Product Details
Description
Product Description
PFI 3 is a potent and selective polybromo 1 (PBRM1) and SMARCA4 inhibitor (Kd values are 48 and 89 nM respectively) that also inhibits SMARCA2. This compoud displays 30-fold selectivity over other sub-family branches. PFI 3 accelerates FRAP recovery in cells at a concentration of 1 μM.Licensing Information
This probe is supplied in conjunction with the Structural Genomics Consortium. For further characterization details, please visit the PFI 3 probe summary on the SGC website.External Portal Information
Chemicalprobes.org is a portal that offers independent guidance on the selection and/or application of small molecules for research. The use of PFI 3 is reviewed on the chemical probes website.
Product Specifications for PFI 3
Molecular Weight
Formula
Storage
Purity
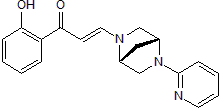
Chemical Name
CAS Number
PubChem ID
InChI Key
SMILES
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Solubility
| Solvent | Max Conc. mg/mL | Max Conc. mM | |
|---|---|---|---|
| Solubility | |||
| DMSO | 32.14 | 100 | |
| Ethanol | 1.61 | 5 with gentle warming |
Preparing Stock Solutions for PFI 3
The following data is based on the product molecular weight 321.37.
Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which all affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 1 mM | 3.11 mL | 15.56 mL | 31.12 mL |
| 5 mM | 0.62 mL | 3.11 mL | 6.22 mL |
| 10 mM | 0.31 mL | 1.56 mL | 3.11 mL |
| 50 mM | 0.06 mL | 0.31 mL | 0.62 mL |
Calculators
Background References
References are publications that support the biological activity of the product. See our Citations tab to view 8 publications citing the usage of this product.
- Gerstenberger Identification of a chemical probe for family VIII bromodomains through optimization of a fragment hit. J.Med.Chem. 2016 PMID: 27115555
- Vangamudi The SMARCA2/4 ATPase domain surpasses the bromodomain as a drug target in SWI/SNF mutant cancers: Insights from cDNA rescue and PFI-3 inhibitor studies Cancer Res. 2015 PMID: 26139243
Product Documents for PFI 3
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for PFI 3
For research use only
Citations for PFI 3
Customer Reviews for PFI 3
There are currently no reviews for this product. Be the first to review PFI 3 and earn rewards!
Have you used PFI 3?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
