Recombinant Cynomolgus Monkey FCAR/CD89 Protein, CF
R&D Systems | Catalog # 9516-FA

Loading...
Key Product Details
- R&D Systems NS0-derived Recombinant Cynomolgus Monkey FCAR/CD89 Protein (9516-FA)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
NS0
Accession Number
Applications
Bioactivity
Loading...
Product Specifications
Source
Mouse myeloma cell line, NS0-derived cynomolgus monkey FCAR/CD89 protein
Gln22-Asn227, with a C-terminal 6-His tag
Gln22-Asn227, with a C-terminal 6-His tag
Purity
>95%, by SDS-PAGE visualized with Silver Staining and quantitative densitometry by Coomassie® Blue Staining.
Endotoxin Level
<0.10 EU per 1 μg of the protein by the LAL method.
N-terminal Sequence Analysis
No reults obtained. Gln 22 inferred from enzymatic pyroglutamate treatment revealing Glu23
Predicted Molecular Mass
24 kDa
SDS-PAGE
33-70 kDa, under reducing conditions.
Activity
Measured by its binding ability in a functional ELISA.
When Human IgA is immobilized at 5 μg/mL, 100 μL/well, the concentration of Recombinant Cynomolgus Monkey FCAR/CD89 that produces 50% of the optimal binding response is 0.250-2.50 μg/mL.
When Human IgA is immobilized at 5 μg/mL, 100 μL/well, the concentration of Recombinant Cynomolgus Monkey FCAR/CD89 that produces 50% of the optimal binding response is 0.250-2.50 μg/mL.
Scientific Data Images for Recombinant Cynomolgus Monkey FCAR/CD89 Protein, CF
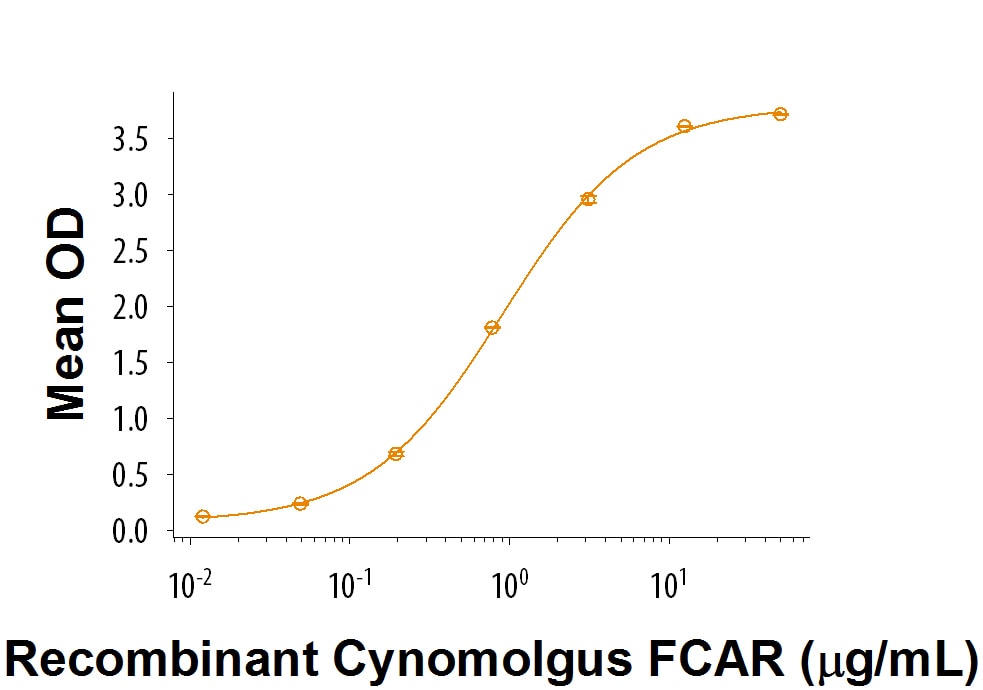
Recombinant Cynomolgus Monkey FCAR/CD89 Protein Bioactivity
When Human IgA is coated at 5 µg/mL (100 μL/well), Recombinant Cynomolgus Monkey FCAR/CD89 (Catalog # 9516-FA) binds with an ED50 of 0.250-2.50 μg/mL.Formulation, Preparation, and Storage
9516-FA
| Formulation | Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. |
| Reconstitution | Reconstitute at 200 μg/mL in PBS.
Loading...
|
| Shipping | The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Calculators
Background: FCAR/CD89
References
- Wines, B. D. and P. M. Hogarth (2006) Tissue Antigens 68:103.
- Otten, M. A. and M. van Egmon (2004) Immunol. Lett. 92:23.
- Montiero, R. C. and J. G. J. van de Winkel (2003) Annu. Rev. Immunol. 21:177.
- Maliszewski, C. R. et al. (1990) J. Exp. Med. 172:1665.
- Launay, P. et al. (1999) J. Biol. Chem. 274:7216.
- Pasquier, B. et al. (2005) Immunity 22:31.
- Shen, L. et al. (2001) Blood 97:205.
- Herr, Y. et al. (2003) Nature 423:614.
- Patry, C. et al. (1996) J. Immunol. 156:4442.
- Togo, S. et al. (2003) FEBS Lett. 535:205.
- van der Boog, P. J. M. et al. (2002) J. Immunol. 168:1252.
- Hamre, R. et al. (2003) Scand. J. Immunol. 57:506.
Long Name
IgA Fc Receptor
Alternate Names
CD89
Gene Symbol
FCAR
UniProt
Additional FCAR/CD89 Products
Product Documents for Recombinant Cynomolgus Monkey FCAR/CD89 Protein, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Cynomolgus Monkey FCAR/CD89 Protein, CF
For research use only
Related Research Areas
Customer Reviews for Recombinant Cynomolgus Monkey FCAR/CD89 Protein, CF
There are currently no reviews for this product. Be the first to review Recombinant Cynomolgus Monkey FCAR/CD89 Protein, CF and earn rewards!
Have you used Recombinant Cynomolgus Monkey FCAR/CD89 Protein, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Loading...
