Recombinant Human Active PIM1 Protein, CF
R&D Systems | Catalog # 4588-KS

Loading...
Key Product Details
- R&D Systems Sf 9 (baculovirus)-derived Recombinant Human Active PIM1 Protein (4588-KS)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
Sf 9 (baculovirus)
Accession Number
Applications
Bioactivity
Loading...
Product Specifications
Source
Spodoptera frugiperda, Sf 9 (baculovirus)-derived human PIM1 protein
Purity
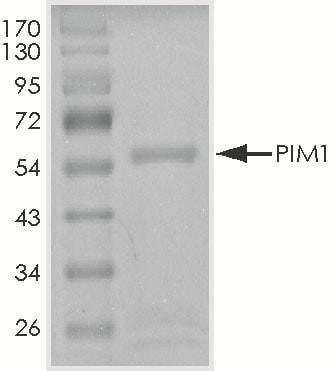
>90%, by SDS-PAGE under reducing conditions and visualized by Colloidal Coomassie® Blue stain at 5 μg per lane.
N-terminal Sequence Analysis
Using an N-terminal GST tag
SDS-PAGE
62 kDa
Activity
The specific activity of PIM1 is typically 269-363 nmol/min/mg using a synthetic peptide substrate.
Scientific Data Images for Recombinant Human Active PIM1 Protein, CF
Recombinant Human Active PIM1 Protein SDS-PAGE.
The approximate molecular weight is 62 kDa and the purity is > 90%.Formulation, Preparation, and Storage
4588-KS
| Formulation | Supplied in 50 mM Tris-HCl, pH 7.5, 150 mM NaCl, 10 mM Glutathione, 0.1 mM EDTA, 0.25 mM DTT, 0.1 mM PMSF, and 25% Glycerol. |
| Shipping | The product is shipped with dry ice or equivalent. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | This product is stable at ≤ ‑70 °C for up to 1 year from the date of receipt. For optimal storage, aliquot into smaller quantities after centrifugation and store at recommended temperature. Avoid repeated freeze-thaw cycles. |
Background: PIM1
References
- Meeker, T.C. et al. (1987) J. Cell. Biochem. 35:105.
- Friedmann, M. et al. (1992) Arch. Biochem. Biophys. 298:594.
Long Name
Proviral Integration Site 1
Alternate Names
EC 2.7.11.1, Oncogene PIM1, PIM, pim-1 kinase 44 kDa isoform, pim-1 oncogene, pim-1 oncogene (proviral integration site 1), proto-oncogene serine/threonine-protein kinase pim-1
Gene Symbol
PIM1
UniProt
Additional PIM1 Products
Product Documents for Recombinant Human Active PIM1 Protein, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Human Active PIM1 Protein, CF
For research use only
Related Research Areas
Customer Reviews for Recombinant Human Active PIM1 Protein, CF
There are currently no reviews for this product. Be the first to review Recombinant Human Active PIM1 Protein, CF and earn rewards!
Have you used Recombinant Human Active PIM1 Protein, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
View specific protocols for Recombinant Human Active PIM1 Protein, CF (4588-KS):
Materials
- Active Kinase - Active PIM1 (0.1 μg/μL) diluted with Kinase Dilution Buffer III to the concentrations indicated in Figure 2. Note: These are suggested working dilutions. Optimal dilutions should be determined by each laboratory for each application.
- Kinase Assay Buffer I - 25 mM MOPS, pH 7.2, 12.5 mM beta -glycerolphosphate, 25 mM MgCl2, 5 mM EGTA, 2 mM EDTA. Add 0.25 mM DTT to the Kinase Assay Buffer prior to use.
- Kinase Dilution Buffer III - Kinase Assay Buffer I diluted at a 1:4 ratio (5X dilution) with 50 ng/μL BSA solution.
- 10 mM ATP Stock Solution - Prepare the ATP Stock Solution by dissolving 55 mg of ATP in 10 mL of Kinase Assay Buffer I. Store 200 μL aliquots at ≤ -20 °C.
- [33P]-ATP Assay Cocktail - Prepare 250 μM [33P]-ATP Assay Cocktail in a designated radioactive work area by combining 150 μL of 10 mM ATP Stock Solution, 100 μL of [33P]-ATP (1 mCi/100 μL), and 5.75 mL of Kinase Assay Buffer I. Store 1 mL aliquots at ≤ -20 °C.
- Substrate - S6K synthetic peptide substrate (KRRRLASLR) diluted in distilled or deionized water to a final concentration of 1 mg/mL.
- Thaw the [33P]-ATP Assay Cocktail in a shielded container in a designated radioactive work area.
- Thaw the Active PIM1, Kinase Assay Buffer I, Substrate, and Kinase Dilution Buffer III on ice.
- In a pre-cooled microfuge tube, add the following reaction components bringing the initial reaction volume up to 20 μL:
a. Diluted Active PIM1: 10 μL
b. Stock solution of Substrate (1 mg/mL): 5 μL
c. Distilled water (2-8 °C): 5 μL - Set up the blank control as outlined in Step 3, excluding the addition of the substrate. Replace the substrate with an equal volume of distilled or deionized water.
- Initiate the reaction with the addition of 5 μL [33P]-ATP Assay Cocktail, bringing the final volume up to 25 μL. Incubate the mixture in a water bath at 30 °C for 15 minutes.
- After the 15 minute incubation, terminate the reaction by spotting 20 μL of the reaction mixture onto individual pre-cut strips of phosphocellulose P81 paper.
- Air dry the pre-cut P81 strip and sequentially wash in a 1% phosphoric acid solution (add 10 mL of phosphoric acid to 990 mL of distilled or deionized water) with constant gentle stirring. It is recommended that the strips be washed a total of three times for approximately 10 minutes each.
- Count the radioactivity (cpm) on the P81 paper in the presence of scintillation fluid in a scintillation counter.
- Determine the corrected cpm by subtracting the blank control value (see Step 4) for each sample and calculate the kinase specific activity as outlined below.
Calculation of [33P]-ATP Specific Activity (SA) (cpm/pmol)
Specific Activity (SA) = cpm for 5 μL [33P]-ATP/pmol of ATP (in 5 μL of a 250 μM ATP stock solution; i.e. 1250 pmol)
Calculation of Kinase Specific Activity (SA) (pmol/minutes/μg or nmol/minutes/mg)
Corrected cpm from reaction / [(SA of 33P-ATP in cpm/pmol) x (Reaction time in minutes) x (Enzyme amount in μg or mg)] x [(Reaction volume) / (Spot Volume)]
Loading...
