Loading...
Key Product Details
- R&D Systems CHO-derived Recombinant Human ADAM15 Protein (9234-AD)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
CHO
Accession Number
Applications
Bioactivity
Loading...
Product Specifications
Source
Chinese Hamster Ovary cell line, CHO-derived human ADAM15 protein
Asp207-Ser693 with a C-terminal 10-His tag
Asp207-Ser693 with a C-terminal 10-His tag
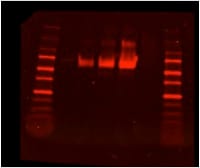
Purity
>95%, by SDS-PAGE visualized with Silver Staining and quantitative densitometry by Coomassie® Blue Staining.
Endotoxin Level
<0.10 EU per 1 μg of the protein by the LAL method.
N-terminal Sequence Analysis
Asp207
Predicted Molecular Mass
53 kDa
SDS-PAGE
67-79 kDa, reducing conditions
Activity
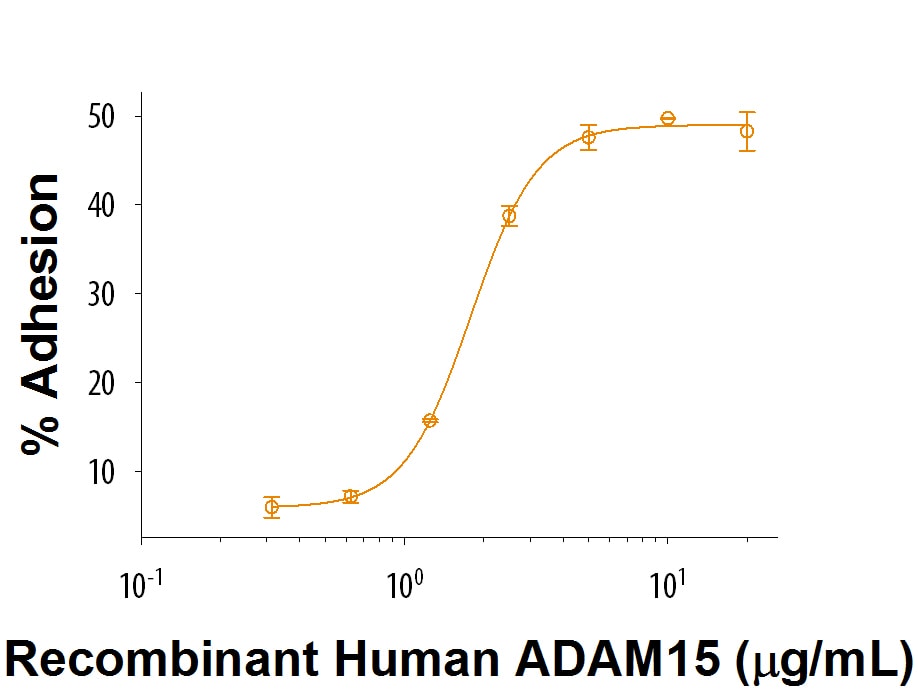
Measured by its ability of the immobilized protein to support the adhesion of Jurkat human acute T cell leukemia cells.
The ED50 for this effect is 0.5-3 μg/mL
The ED50 for this effect is 0.5-3 μg/mL
Scientific Data Images for Recombinant Human ADAM15 Protein, CF
Recombinant Human ADAM15 Protein Bioactivity
Immobilized Recombinant Human ADAM15 (Catalog # 9234-AD) supports the adhesion of Jurkat human acute T cell leukemia cells. The ED50 for this effect is 0.5-3 μg/mL.Formulation, Preparation, and Storage
9234-AD
| Formulation | Lyophilized from a 0.2 μm filtered solution in MES and NaCl. |
| Reconstitution | Reconstitute at 500 μg/mL in water.
Loading...
|
| Shipping | The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Calculators
Background: ADAM15
N-Cadherin, E-Cadherin, MICB, CD23/Fc epsilon RII, and FGF R2 (IIb) (6, 7, 14-16).
References
- Edwards, D.R. et al. (2008) Mol. Aspects Med. 29:258.
- Kratzschmar, J. et al. (1996) J. Biol. Chem. 271:4593.
- Kleino, I. et al. (2009) J. Cell. Biochem. 108:877.
- Kleino, I. et al. (2007) BMC Mol. Biol. 8:90.
- Mosnier, J.F. et al. (2006) Lab. Invest. 86:1064.
- Najy, A.J. et al. (2008) Cancer Res. 68:1092.
- Duan, X. et al. (2013) Mol. Med. Rep. 7:991.
- Dong, D.D. et al. (2015) Oncol. Rep. 34:2451.
- Pasten-Hidalgo, K. et al. (2008) Reproduction 136:41.
- Pasten, K. et al. (2014) Reproduction 148:623.
- Sun, C. et al. (2010) Cardiovasc. Res. 87:348.
- Charrier, L. et al. (2007) J. Biol. Chem. 282:16948.
- Zhang, X.P. et al. (1998) J. Biol. Chem. 273:7345.
- Najy, A.J. et al. (2008) J. Biol. Chem. 283:18393.
- Fourie, A.M. et al. (2003) J. Biol. Chem. 278:30469.
- Maretzky, T. et al. (2009) Biochem. J. 420:105.
Long Name
A Disintegrin and Metalloprotease-like Domain 15
Alternate Names
MDC15, Metargidin
Gene Symbol
ADAM15
UniProt
Additional ADAM15 Products
Product Documents for Recombinant Human ADAM15 Protein, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Human ADAM15 Protein, CF
For research use only
Related Research Areas
Customer Reviews for Recombinant Human ADAM15 Protein, CF (1)
4 out of 5
1 Customer Rating
Have you used Recombinant Human ADAM15 Protein, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Verified Customer | Posted 09/27/2017
There are no reviews that match your criteria.
Loading...