Aldehyde dehydrogenases (ALDHs) are NAD(P)+-dependent enzymes that detoxify aldehydes by oxidizing them to carboxylic acids. Nineteen ALDHs are present in humans, expressed in a variety of organelles and having different substrate preferences (1). Human ALDH1A2 is a NAD-dependent, cytosolic member of the ALDH1A subfamily class 1 that forms active tetramers that synthesize retinoic acid from retinal. Each monomer has three functional regions: a catalytic domain containing the active site cysteine, an NAD-binding domain, and the oligomerization domain (2, 3). ALDH1A2 exhibits the highest substrate specificity and catalytic efficiency for retinal oxidation to retinoic acid (RA) (4) and thus plays an important role in regulation of RA production and signaling. ALDH1A2 null mice are embryonic lethal (5) and mutations in ALDH1A2 are associated with pathological conditions such as osteoarthris (6-8). Additionally, ALDH1A2 is suggested to play a role in several cancers. ALDH1A2 was a suggested tumor marker (9, 10) and predictor of prostate cancer relapse (11). Abnormally low levels of ALDH1A2 has been observed in several cancers including breast (12), squamous cell carcinoma of the head and neck (13), and specifically in high grade ovarian cancer, suggesting high expression may be associated with favorable prognosis (14). Overexpression of ALDH1A2 in cancer lines resulted in decreased proliferation and migration (14) supporting that ALDH1A2 plays a tumor suppressor role. In contrast, ALDH1A2 is reported to exhibit high expression correlated with worse overall survival in non-small-cell lung cancer (15) and chemoresistant cancer stem cells in neuroblastoma (16, 17). Pharmacologically specific inhibitors and activators of ALDH1A2 are of interest due to its implied role in a variety of diseases and proliferation and drug resistance (1, 3, 15, 18).
Recombinant Human ALDH1A2 His-tag, CF
R&D Systems | Catalog # 10132-DH

Key Product Details
- R&D Systems E. coli-derived Recombinant Human ALDH1A2 His-tag (10132-DH)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
Accession Number
Applications
Product Specifications
Source
Thr2-Ser518
with an N-terminal Met and 6-His tag
Purity
Endotoxin Level
N-terminal Sequence Analysis
Predicted Molecular Mass
SDS-PAGE
Activity
The specific activity is >150 pmol/min/μg, as measured under the described conditions.
Scientific Data Images for Recombinant Human ALDH1A2 His-tag, CF
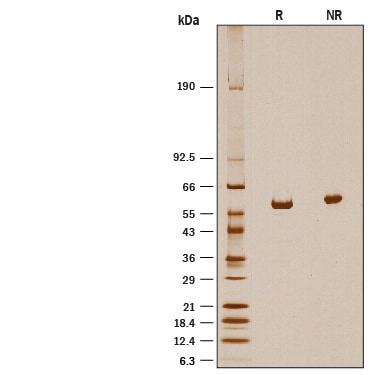
Recombinant Human ALDH1A2 His-tag SDS-PAGE
1 μg/lane of Recombinant Human ALDH1A2 His-tag was resolved with SDS-PAGE under reducing (R) and non-reducing (NR) conditions and visualized by silver staining, showing a band at 58 kDa under reducing conditions.Formulation, Preparation, and Storage
10132-DH
| Formulation | Supplied as a 0.2 μm filtered solution in Tris, NaCl, Glycerol and DTT. |
| Shipping | The product is shipped with dry ice or equivalent. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Background: ALDH1A2
References
- Koppaka, V. et al. (2012) Pharmacol. Rev. 64:520.
- Perez-Miller, S. J. and T. D. Hurley (2003) Biochemistry. 42:7100.
- Chen, Y. et al. (2018) ACS Chem. Biol. 13:582.
- Duester, G. (2001) Chem. Biol. Interact. 130:469.
- Niedereither, K. et al. (1999) Nat. Genet. 21:444.
- Pavan, M. et al. (2009) B.M.C. Med. Genet. 10:113.
- Styrkarsdottir, U. et al. (2014) Nat. Genet. 46:498.
- Shepherd, C. et. al. (2018) Arthritis Rheumatol. 70:1577.
- Kim, H. et al. (2005) Cancer Res. 65:8118.
- Touma, S. E. et al. (2009) Biochem. Pharmacol. 78:1127.
- Nim, H. T. et al. (2017) Front. Oncol. 7:30.
- Mira, Y. L. R. et al. (2000) J. Cell Physiol. 185:302.
- Seidensaal, K. et al. (2015) Mol. Cancer 14:204.
- Wang, Y. et al. (2018) Onco. Targets Ther. 31:599.
- You, Q. et al. (2015) Drug Des. Devel. Ther. 9:5087.
- Hartomo, T. B. et al. (2015) Int. J. Oncol. 46:1089.
- Singh, S. et al. (2015) Adv. Exp. Med. Biol. 815:281.
- Moreb, J. S. et al. (2012) Chem. Biol. Interact. 195:52.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional ALDH1A2 Products
Product Documents for Recombinant Human ALDH1A2 His-tag, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Human ALDH1A2 His-tag, CF
For research use only
Related Research Areas
Customer Reviews for Recombinant Human ALDH1A2 His-tag, CF
There are currently no reviews for this product. Be the first to review Recombinant Human ALDH1A2 His-tag, CF and earn rewards!
Have you used Recombinant Human ALDH1A2 His-tag, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
View specific protocols for Recombinant Human ALDH1A2 His-tag, CF (10132-DH):
- Assay Buffer: 50 mM Tris, 100 mM KCl, 2 mM DTT, pH 8.5
- Recombinant Human ALDH1A2 (rhALDH1A2) (Catalog # 10132-DH)
- beta -Nicotinamide adenine dinucleotide ( beta -NAD) (Sigma, Catalog # N6522), 100 mM stock in deionized water
- 4-Nitrobenzaldehyde (4-NBA) (Sigma, Catalog # 72800), 200 mM stock in DMSO
- 96-well Clear Plate (Catalog # DY990)
- Plate Reader (Model: SpectraMax Plus by Molecular Devices) or equivalent
- Dilute rhALDH1A2 to 10 µg/mL in Assay Buffer.
- Prepare Substrate Mixture containing 2 mM beta -NAD and 2 mM 4-NBA in Assay Buffer.
- Load 50 µL of 10 µg/mL rhALDH1A2 into the plate, and start the reaction by adding 50 µL of Substrate Mixture. Include a Substrate Blank containing 50 µL of Assay Buffer and 50 µL of Substrate Mixture.
- Read plate at 340 nm (absorbance) in kinetic mode for 5 minutes.
- Calculate specific activity:
Specific Activity (pmol/min/µg) = | Adjusted Vmax* (OD/min) x well volume (L) x 1012 pmol/mol |
| ext. coeff** (M-1cm-1) x path corr.*** (cm) x amount of enzyme (µg) |
*Adjusted for Substrate Blank
**Using the extinction coefficient 6220 M-1cm-1
***Using the path correction 0.320 cm
Note: the output of many spectrophotometers is in mOD
Per Well:
- rhALDH1A2: 0.5 µg
- beta -NAD: 1 mM
- 4-NBA: 1 mM
