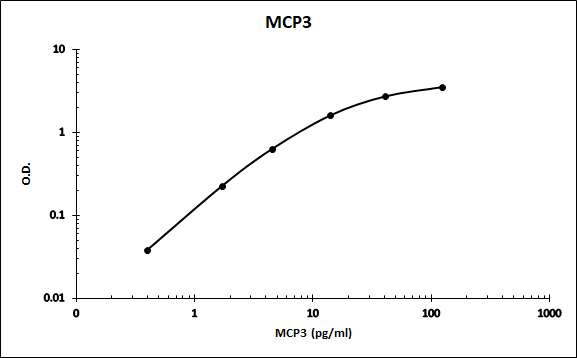
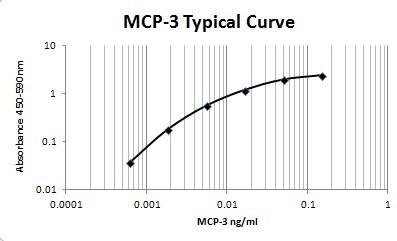
MCP-2 and CCL7 are two monocyte chemotactic proteins produced by human MG-63 osteosarcoma cells. Both MCP-2 and CCL7 are members of the C-C family of chemokines and share 62% and 71% amino acid sequence identity, respectively, with MCP-1. CCL7 also shares 58% amino acid identity with MCP-2.
CCL7 cDNA encodes a 99 amino acid residue precursor protein from which the N-terminal 23 amino acid residues are cleaved to generate the 76 amino acid residue mature CCL7. Mature CCL7 contains a potential N-linked and several possible O-linked glycosylation sites.
Similarly to other C-C chemokines, all three MCP proteins are monocyte chemoattractants. In addition, the three MCPs can chemoattract activated NK cells as well as CD4+ and CD8+ T lymphocytes. All three cytokines have also been shown to attract eosinophils and induce histamine secretion from basophils.