Recombinant Human CD9-LEL Fc Chimera Protein, CF
R&D Systems | Catalog # 10015-CD

Loading...
Key Product Details
- R&D Systems CHO-derived Recombinant Human CD9-LEL Fc Chimera Protein (10015-CD)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
CHO
Accession Number
Structure / Form
Disulfide-linked homodimer
Applications
Bioactivity
Loading...
Product Specifications
Source
Chinese Hamster Ovary cell line, CHO-derived human CD9 protein
| Human IgG1 MD + (Pro100-Lys330) |
IEGR | Human CD9-LEL (Ser112-Ile195) Accession # P21926 |
| N-terminus | C-terminus |
Purity
>95%, by SDS-PAGE visualized with Silver Staining and quantitative densitometry by Coomassie® Blue Staining.
Endotoxin Level
<0.10 EU per 1 μg of the protein by the LAL method.
N-terminal Sequence Analysis
MD + Pro100
Predicted Molecular Mass
36.3 kDa
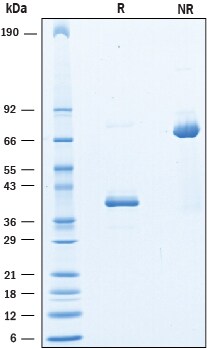
SDS-PAGE
38-42 kDa, reducing conditions
Activity
Measured by its binding ability in a functional ELISA.
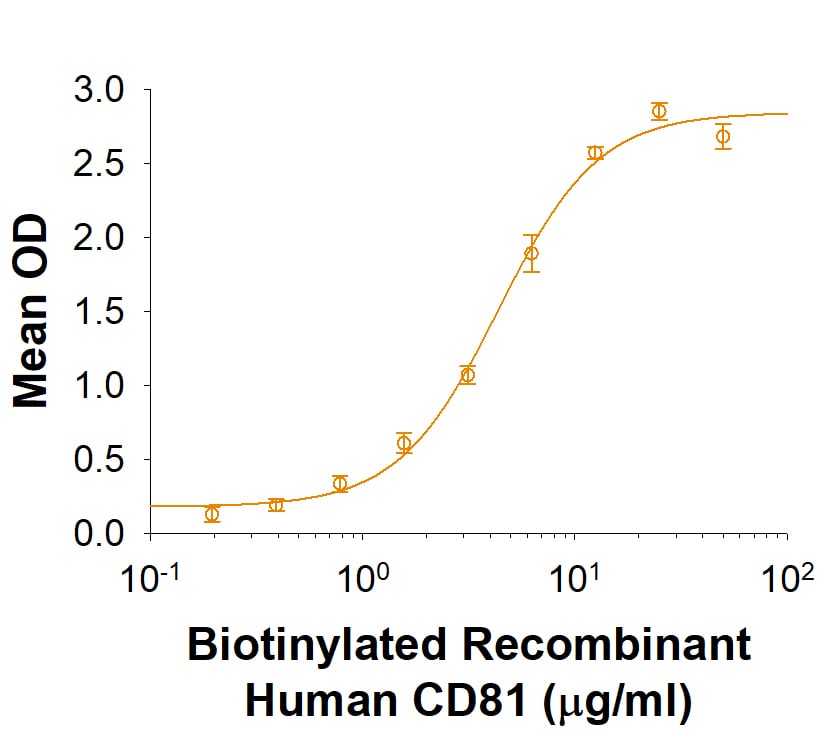
When Recombinant Human CD9 Fc Chimera is immobilized at 1 µg/mL (100 µL/well), Biotinylated Recombinant Human CD81 Fc Chimera binds with an ED50 of 2-10 μg/mL.
When Recombinant Human CD9 Fc Chimera is immobilized at 1 µg/mL (100 µL/well), Biotinylated Recombinant Human CD81 Fc Chimera binds with an ED50 of 2-10 μg/mL.
Scientific Data Images for Recombinant Human CD9-LEL Fc Chimera Protein, CF
Recombinant Human CD9-LEL Fc Chimera Protein Binding Activity
When Recombinant Human CD9 Fc Chimera (Catalog # 10015-CD) is immobilized at 1 µg/mL, Biotinylated Recombinant CD81 Fc Chimera binds with an ED50 of 2-10 μg/mL.Recombinant Human CD9-LEL Fc Chimera Protein SDS-PAGE
2 μg/lane of Recombinant Human CD9 was resolved with SDS-PAGE under reducing (R) and non-reducing (NR) conditions and visualized by Coomassie® Blue staining, showing bands at 38-42 kDa and 75-85 kDa, respectively.Formulation, Preparation, and Storage
10015-CD
| Formulation | Lyophilized from a 0.2 μm filtered solution in PBS. |
| Reconstitution | Reconstitute at 500 μg/mL in PBS. |
| Shipping | The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Calculators
Background: CD9
References
- Shi, W. et al. (2000) J. Cell Biol. 148:591.
- Hemler, M. (2003) Annu Rev Cell Biol. 19:397.
- Hulme, R. et al. (2014) PLoS One 9:e116289.
- Stipp, C. et al. (2003) Trends Biochem Sci. 28:106.
- Barreiro, O. et al. (2005) Blood 105:2852.
- Ventress, J. et al. (2016) PLoS One 11:e0160387.
- Rubinstein, E. (2011) Biochem Soc Trans. 39:501.
- Anzai, N. et al. (2002) Blood 99:4413.
- Radford, K. et al. (1996) Biochem. Biophys. Res. Commun. 222:13.
- Lozahic, S. et al. (2000) Eur. J. Immunol. 30:900.
- Park, K. et al. (2000) Mol. Hum. Reprod. 6:252.
- Charrin, S. et al. (2001) J. Biol. Chem. 276:14329.
- Tachibana, I. et al. (1997) J. Biol. Chem. 272:29181.
- Zhu, G. et al. (2002) Development 129:1995.
- Green, L. et al. (2011) Infect Immun. 79:2241.
- Powner, D. et al. (2011) Biochem. Soc. Trans. 39:563.
- Detchokul, S. et al. (2014) British Journal of Pharmacology 171:5462.
- Reyes, R. et al. (2018) Front. Immunol. 9:863.
- Slupsky, J. et al. (1989) J Biol chem. 264:12289.
- Ishibashi, T. et al. (2004) J. Neuroscience 24:96.
- Kobayashi, H. et al. (2004) Clin Exp Immunol. 137:101.
Alternate Names
CD9, DRAP-27, MIC3, p24, TSPAN29
Gene Symbol
CD9
UniProt
Additional CD9 Products
Product Documents for Recombinant Human CD9-LEL Fc Chimera Protein, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Human CD9-LEL Fc Chimera Protein, CF
For research use only
Related Research Areas
Customer Reviews for Recombinant Human CD9-LEL Fc Chimera Protein, CF
There are currently no reviews for this product. Be the first to review Recombinant Human CD9-LEL Fc Chimera Protein, CF and earn rewards!
Have you used Recombinant Human CD9-LEL Fc Chimera Protein, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Loading...