The coronavirus Spike protein receptor binding domain (RBD) resides within the S1 subunit and is responsible for binding to host cell receptors and initiating viral infection. Past coronaviruses SARS and MERS along with the global pandemic caused by SARS-CoV-2 have sparked great interest and scientific discovery leading to new vaccines and drug development. At the heart of coronavirus biology lies the Spike protein and its receptor binding domain. The Spike RBD is a 26 kDa domain consisting of a twisted five-stranded antiparallel beta sheet and sits at the apex of each Spike protein monomer. The Spike RBD is flexible thanks to a hinge region that allows for conformational changes that expose (up or open conformation) or hide (down or closed conformation) its receptor contacts. For SARS-CoV-2, the Spike RBD recognizes and tightly binds to the human receptor ACE-2. In the closely related MERS coronavirus, human DPPIV/CD26 acts as the receptor.
SARS-CoV-2 Variant Inhibitor Screening Kit
R&D Systems | Catalog # VANC00
Omicron Variant Now Availble, Catalog # VANC00B!

Product Specifications
Detection Method
Specificity
Label
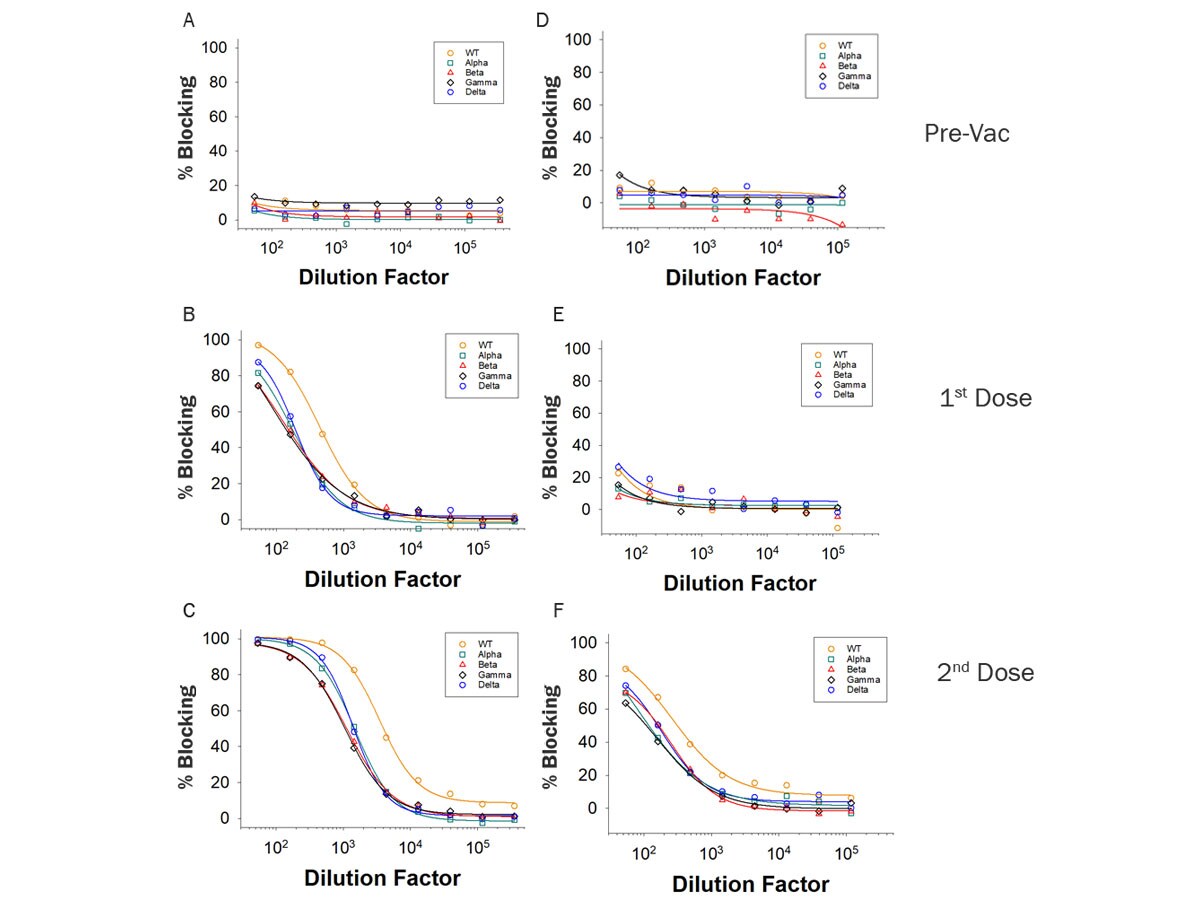
Scientific Data Images for SARS-CoV-2 Variant Inhibitor Screening Kit
Preparation and Storage
Shipping
Stability & Storage
Background: Spike RBD
Long Name
Gene Symbol
Additional Spike RBD Products
Product Documents for SARS-CoV-2 Variant Inhibitor Screening Kit
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for SARS-CoV-2 Variant Inhibitor Screening Kit
For research use only
Related Research Areas
Customer Reviews for SARS-CoV-2 Variant Inhibitor Screening Kit
There are currently no reviews for this product. Be the first to review SARS-CoV-2 Variant Inhibitor Screening Kit and earn rewards!
Have you used SARS-CoV-2 Variant Inhibitor Screening Kit?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
FAQs for SARS-CoV-2 Variant Inhibitor Screening Kit
-
Q: Is it okay to modify the plate layout in the kit product datasheet?
A: Yes, a plate layout is provided in the kit insert as an example, and investigators can modify the plate layout to suit specific needs. Spike RBD WT or variant proteins are supplied at higher amounts to provide flexibility for investigators to use each protein of interest as desired.
-
Q: The SARS-CoV-2 variants that I am interested in are not listed in the kit insert. Is there an option to obtain a kit that contains other variants?
A:
Yes; to discuss other available options, please contact our Technical Support by email techsupport@bio-techne.com or by phone Toll Free USA, Canada: 1-800-343-7475
Phone (International): 1-800-343-7475.
-
Q: What are possible reasons for low assay signal?
A: Low assay signal may be due to non-optimal preparation of Recombinant SARS2 Spike RBD proteins or Biotinylated Human ACE-2. Be sure to review the product insert and follow reconstitution and dilution instructions for each lyophilized reagent. Low signal may also be caused by insufficient incubation time for color reaction. Color development for the Covid-19 Neutralization assay requires 15 to 25 minutes.
-
Q: What can cause high background?
A: Problem of high background may be due to insufficient washing or extended incubation time with detection reagents, among other factors.
-
Q: What could cause high variability between sample duplicates?
A: The two main reasons for high variability in this assay are non-optimal pipetting technique and samples not well mixed. Ensure a consistent and accurate pipetting method. Mix sample thoroughly and dispense accurately. Change pipette tips between samples. Ensure that your pipettes are calibrated.
-
Q: Why are Sample Dilution Plates provided in the kit?
A: The Sample Dilution Plates have reduced protein binding to minimize loss of sample during the dilution and pre-incubation steps, and the plates are provided in the kit for convenience of use. Diluted samples prepared in a dilution plate can be transferred to a 96-well Assay Plate in an efficient manner minimizing chance for assay drift.
-
Q: Is it okay to modify the plate layout in the kit product datasheet?
A: Yes, a plate layout is provided in the kit insert as an example, and investigators can modify the plate layout to suit specific needs. Spike RBD WT or variant proteins are supplied at higher amounts to provide flexibility for investigators to use each protein of interest as desired.
-
Q: The SARS-CoV-2 variants that I am interested in are not listed in the kit insert. Is there an option to obtain a kit that contains other variants?
A:
Yes; to discuss other available options, please contact our Technical Support by email techsupport@bio-techne.com or by phone Toll Free USA, Canada: 1-800-343-7475
Phone (International): 1-800-343-7475.
-
Q: What are possible reasons for low assay signal?
A: Low assay signal may be due to non-optimal preparation of Recombinant SARS2 Spike RBD proteins or Biotinylated Human ACE-2. Be sure to review the product insert and follow reconstitution and dilution instructions for each lyophilized reagent. Low signal may also be caused by insufficient incubation time for color reaction. Color development for the Covid-19 Neutralization assay requires 15 to 25 minutes.
-
Q: What can cause high background?
A: Problem of high background may be due to insufficient washing or extended incubation time with detection reagents, among other factors.
-
Q: What could cause high variability between sample duplicates?
A: The two main reasons for high variability in this assay are non-optimal pipetting technique and samples not well mixed. Ensure a consistent and accurate pipetting method. Mix sample thoroughly and dispense accurately. Change pipette tips between samples. Ensure that your pipettes are calibrated.
-
Q: Why are Sample Dilution Plates provided in the kit?
A: The Sample Dilution Plates have reduced protein binding to minimize loss of sample during the dilution and pre-incubation steps, and the plates are provided in the kit for convenience of use. Diluted samples prepared in a dilution plate can be transferred to a 96-well Assay Plate in an efficient manner minimizing chance for assay drift.
-
Q: Is it okay to modify the plate layout in the kit product datasheet?
A: Yes, a plate layout is provided in the kit insert as an example, and investigators can modify the plate layout to suit specific needs. Spike RBD WT or variant proteins are supplied at higher amounts to provide flexibility for investigators to use each protein of interest as desired.
-
Q: The SARS-CoV-2 variants that I am interested in are not listed in the kit insert. Is there an option to obtain a kit that contains other variants?
A:
Yes; to discuss other available options, please contact our Technical Support by email techsupport@bio-techne.com or by phone Toll Free USA, Canada: 1-800-343-7475
Phone (International): 1-800-343-7475.
-
Q: What are possible reasons for low assay signal?
A: Low assay signal may be due to non-optimal preparation of Recombinant SARS2 Spike RBD proteins or Biotinylated Human ACE-2. Be sure to review the product insert and follow reconstitution and dilution instructions for each lyophilized reagent. Low signal may also be caused by insufficient incubation time for color reaction. Color development for the Covid-19 Neutralization assay requires 15 to 25 minutes.
-
Q: What can cause high background?
A: Problem of high background may be due to insufficient washing or extended incubation time with detection reagents, among other factors.
-
Q: What could cause high variability between sample duplicates?
A: The two main reasons for high variability in this assay are non-optimal pipetting technique and samples not well mixed. Ensure a consistent and accurate pipetting method. Mix sample thoroughly and dispense accurately. Change pipette tips between samples. Ensure that your pipettes are calibrated.
-
Q: Why are Sample Dilution Plates provided in the kit?
A: The Sample Dilution Plates have reduced protein binding to minimize loss of sample during the dilution and pre-incubation steps, and the plates are provided in the kit for convenience of use. Diluted samples prepared in a dilution plate can be transferred to a 96-well Assay Plate in an efficient manner minimizing chance for assay drift.
-
Q: Is it okay to modify the plate layout in the kit product datasheet?
A: Yes, a plate layout is provided in the kit insert as an example, and investigators can modify the plate layout to suit specific needs. Spike RBD WT or variant proteins are supplied at higher amounts to provide flexibility for investigators to use each protein of interest as desired.
-
Q: The SARS-CoV-2 variants that I am interested in are not listed in the kit insert. Is there an option to obtain a kit that contains other variants?
A:
Yes; to discuss other available options, please contact our Technical Support by email techsupport@bio-techne.com or by phone Toll Free USA, Canada: 1-800-343-7475
Phone (International): 1-800-343-7475.
-
Q: What are possible reasons for low assay signal?
A: Low assay signal may be due to non-optimal preparation of Recombinant SARS2 Spike RBD proteins or Biotinylated Human ACE-2. Be sure to review the product insert and follow reconstitution and dilution instructions for each lyophilized reagent. Low signal may also be caused by insufficient incubation time for color reaction. Color development for the Covid-19 Neutralization assay requires 15 to 25 minutes.
-
Q: What can cause high background?
A: Problem of high background may be due to insufficient washing or extended incubation time with detection reagents, among other factors.
-
Q: What could cause high variability between sample duplicates?
A: The two main reasons for high variability in this assay are non-optimal pipetting technique and samples not well mixed. Ensure a consistent and accurate pipetting method. Mix sample thoroughly and dispense accurately. Change pipette tips between samples. Ensure that your pipettes are calibrated.
-
Q: Why are Sample Dilution Plates provided in the kit?
A: The Sample Dilution Plates have reduced protein binding to minimize loss of sample during the dilution and pre-incubation steps, and the plates are provided in the kit for convenience of use. Diluted samples prepared in a dilution plate can be transferred to a 96-well Assay Plate in an efficient manner minimizing chance for assay drift.
-
Q: Is it okay to modify the plate layout in the kit product datasheet?
A: Yes, a plate layout is provided in the kit insert as an example, and investigators can modify the plate layout to suit specific needs. Spike RBD WT or variant proteins are supplied at higher amounts to provide flexibility for investigators to use each protein of interest as desired.
-
Q: The SARS-CoV-2 variants that I am interested in are not listed in the kit insert. Is there an option to obtain a kit that contains other variants?
A:
Yes; to discuss other available options, please contact our Technical Support by email techsupport@bio-techne.com or by phone Toll Free USA, Canada: 1-800-343-7475
Phone (International): 1-800-343-7475.
-
Q: What are possible reasons for low assay signal?
A: Low assay signal may be due to non-optimal preparation of Recombinant SARS2 Spike RBD proteins or Biotinylated Human ACE-2. Be sure to review the product insert and follow reconstitution and dilution instructions for each lyophilized reagent. Low signal may also be caused by insufficient incubation time for color reaction. Color development for the Covid-19 Neutralization assay requires 15 to 25 minutes.
-
Q: What can cause high background?
A: Problem of high background may be due to insufficient washing or extended incubation time with detection reagents, among other factors.
-
Q: What could cause high variability between sample duplicates?
A: The two main reasons for high variability in this assay are non-optimal pipetting technique and samples not well mixed. Ensure a consistent and accurate pipetting method. Mix sample thoroughly and dispense accurately. Change pipette tips between samples. Ensure that your pipettes are calibrated.
-
Q: Why are Sample Dilution Plates provided in the kit?
A: The Sample Dilution Plates have reduced protein binding to minimize loss of sample during the dilution and pre-incubation steps, and the plates are provided in the kit for convenience of use. Diluted samples prepared in a dilution plate can be transferred to a 96-well Assay Plate in an efficient manner minimizing chance for assay drift.
-
Q: Is it okay to modify the plate layout in the kit product datasheet?
A: Yes, a plate layout is provided in the kit insert as an example, and investigators can modify the plate layout to suit specific needs. Spike RBD WT or variant proteins are supplied at higher amounts to provide flexibility for investigators to use each protein of interest as desired.
-
Q: The SARS-CoV-2 variants that I am interested in are not listed in the kit insert. Is there an option to obtain a kit that contains other variants?
A:
Yes; to discuss other available options, please contact our Technical Support by email techsupport@bio-techne.com or by phone Toll Free USA, Canada: 1-800-343-7475
Phone (International): 1-800-343-7475.
-
Q: What are possible reasons for low assay signal?
A: Low assay signal may be due to non-optimal preparation of Recombinant SARS2 Spike RBD proteins or Biotinylated Human ACE-2. Be sure to review the product insert and follow reconstitution and dilution instructions for each lyophilized reagent. Low signal may also be caused by insufficient incubation time for color reaction. Color development for the Covid-19 Neutralization assay requires 15 to 25 minutes.
-
Q: What can cause high background?
A: Problem of high background may be due to insufficient washing or extended incubation time with detection reagents, among other factors.
-
Q: What could cause high variability between sample duplicates?
A: The two main reasons for high variability in this assay are non-optimal pipetting technique and samples not well mixed. Ensure a consistent and accurate pipetting method. Mix sample thoroughly and dispense accurately. Change pipette tips between samples. Ensure that your pipettes are calibrated.
-
Q: Why are Sample Dilution Plates provided in the kit?
A: The Sample Dilution Plates have reduced protein binding to minimize loss of sample during the dilution and pre-incubation steps, and the plates are provided in the kit for convenience of use. Diluted samples prepared in a dilution plate can be transferred to a 96-well Assay Plate in an efficient manner minimizing chance for assay drift.