The coronavirus Spike protein receptor binding domain (RBD) resides within the S1 subunit and is responsible for binding to host cell receptors and initiating viral infection. Past coronaviruses SARS and MERS along with the global pandemic caused by SARS-CoV-2 have sparked great interest and scientific discovery leading to new vaccines and drug development. At the heart of coronavirus biology lies the Spike protein and its receptor binding domain. The Spike RBD is a 26 kDa domain consisting of a twisted five-stranded antiparallel beta sheet and sits at the apex of each Spike protein monomer. The Spike RBD is flexible thanks to a hinge region that allows for conformational changes that expose (up or open conformation) or hide (down or closed conformation) its receptor contacts. For SARS-CoV-2, the Spike RBD recognizes and tightly binds to the human receptor ACE-2. In the closely related MERS coronavirus, human DPPIV/CD26 acts as the receptor.
SARS-CoV-2 Variant Inhibitor Screening Kit
R&D Systems | Catalog # VANC00B
COVID-19
Now with Omicron and Delta Variant!
Now with Omicron and Delta Variant!

Loading...
Loading...
Product Specifications
Detection Method
Colorimetric ELISA - 450nm (TMB)
Specificity
Label
HRP
Scientific Data Images for SARS-CoV-2 Variant Inhibitor Screening Kit
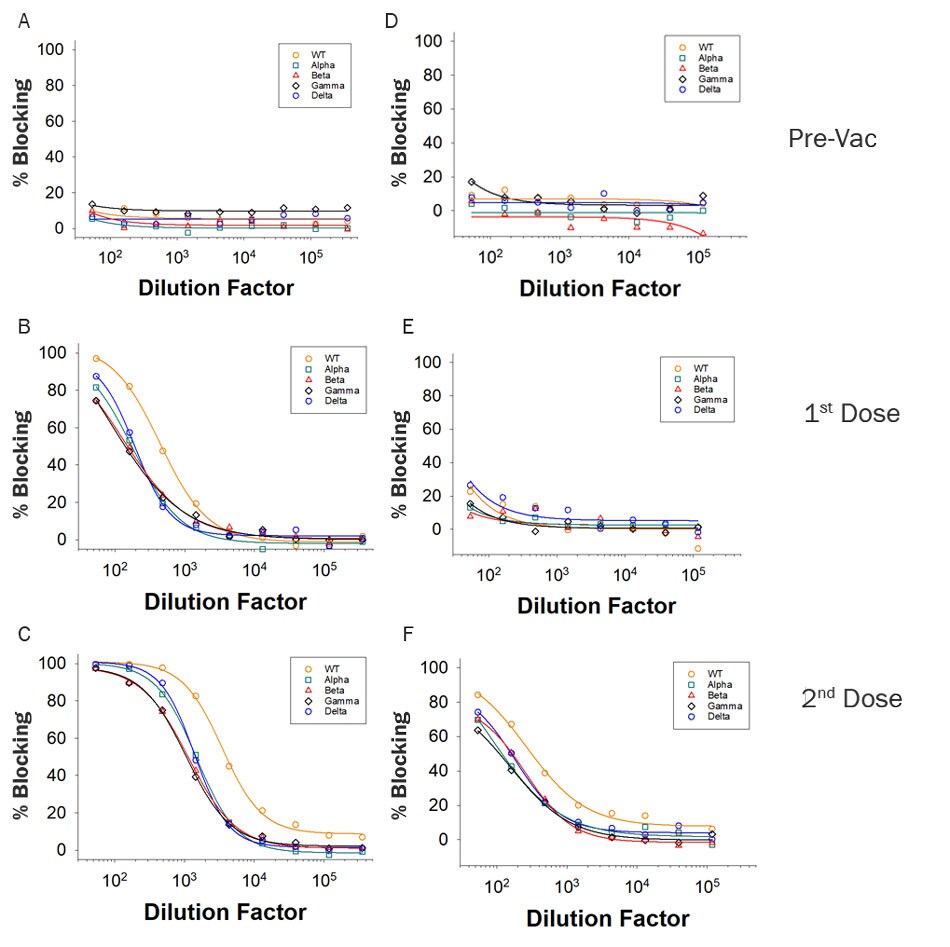
Figure 1: Example data using vaccinated donor samples.
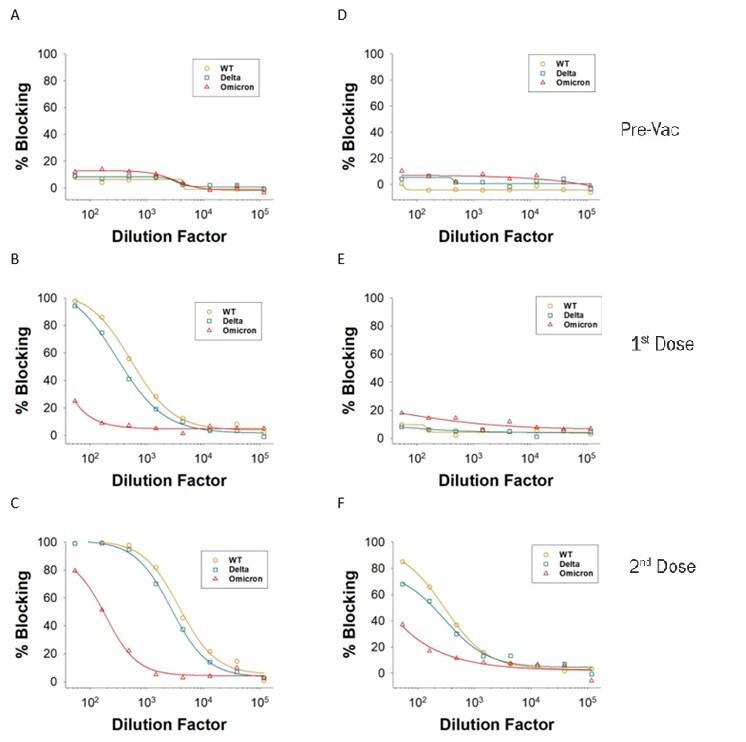
Serological samples were collected from two donors over a time course consisting Pre-Vaccination (Pre-Vac), or after receiving the 1st Dose or 2nd Dose of the Moderna Vaccine. Donor 1 (A-C) and Donor 2 (D-F) had different blocking antibody levels after the first and second doses. In addition, increasing amounts of blocking antibodies were present at higher samples Dilution Factors after each dose. The differences in blocking efficiency of ACE-2 association between the WT, Alpha, Beta, Gamma, or Delta variant Spike RBD was measured.Figure 2: Example data using vaccinated donor samples Using the Wild Type or the Delta and Omicron Variants.
Plasma samples were collected from two donors over a time course consisting of Pre-Vaccination (Pre-Vac), or after receiving the 1st Dose or 2nd Dose of the Moderna Vaccine. Donor 1 (A-C) and Donor 2 (D-F) had different blocking antibody levels after the first and second doses. In addition, increasing amounts of blocking antibodies were present at higher samples Dilution Factors after each dose. The differences in blocking efficiency of ACE-2 association between the WT, Delta, or Omicron variant Spike RBD was measured.Preparation and Storage
Shipping
The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below.
Stability & Storage
Store the unopened product at 2 - 8 °C. Do not use past expiration date.
Background: Spike RBD
Long Name
Spike Receptor Binding Domain
Gene Symbol
S
Additional Spike RBD Products
Product Documents for SARS-CoV-2 Variant Inhibitor Screening Kit
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for SARS-CoV-2 Variant Inhibitor Screening Kit
For research use only
Related Research Areas
Citations for SARS-CoV-2 Variant Inhibitor Screening Kit
Customer Reviews for SARS-CoV-2 Variant Inhibitor Screening Kit
There are currently no reviews for this product. Be the first to review SARS-CoV-2 Variant Inhibitor Screening Kit and earn rewards!
Have you used SARS-CoV-2 Variant Inhibitor Screening Kit?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Loading...