Maurice Instrument Applications
Maurice and MauriceFlex
Maurice and MauriceFlex simplify the analysis of charge, purity, identity, and other CQAs with fully automated CE‑SDS, icIEF, and icIEF fractionation workflows. These systems automate workflows and reduce manual burden while improving data reproducibility and speed, accelerating development from early research to commercial manufacturing.

icIEF- Imaged Charge Isoelectric Focusing
Enhance charge variant profiling with high‑precision icIEF. Use Maurice instruments to generate consistent, high‑resolution isoelectric focusing data for monoclonal antibodies and complex biologics. Automated icIEF improves accuracy compared to IEX and strengthens charge heterogeneity assessment across development stages.
icIEF for charge variant and charge heterogeneity analysis
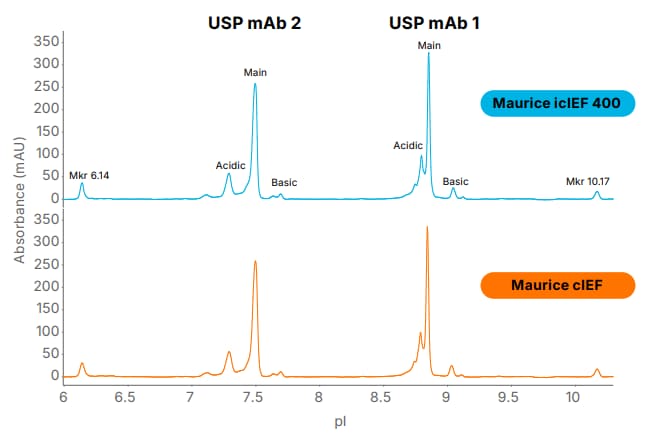
Figure 1: Analysis of USP mAb 1 and USP mAb 2 with the Maurice icIEF 400 and Maurice cIEF cartridges. The profiles show similar resolution for charge variants and near-identical peak distribution, confirming comparable performance.
icIEF-Based Protein Fractionation for In-Depth Characterization
Get a deeper structural understanding of your molecules. Use MauriceFlex to fractionate charge variants with high‑resolution icIEF and collect LC‑MS‑ready fractions without buffer exchange or added sample prep. This lets you characterize intact mass, reduced mass, peptide mapping, binding interactions, and aggregates with a simplified workflow.
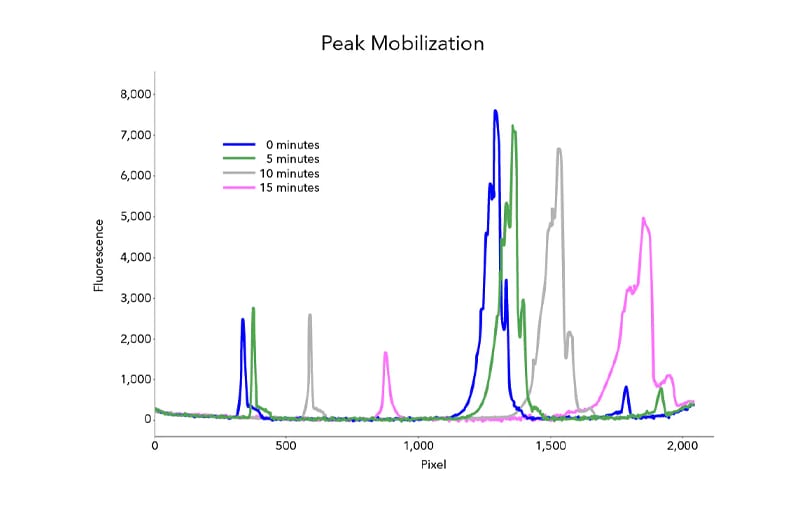
Figure 2: Peak mobilization of charge isoforms of a typical monoclonal antibody, achieved on the MauriceFlex system.
CE-SDS for Protein Purity, Identity, and Size Analysis
Speed up purity and identity analysis with automated CE‑SDS. Use the CE-SDS application on Maurice and MauriceFlex to replace manual SDS‑PAGE with fully automated, high‑quality, quantitative workflows. Run both Turbo CE‑SDS and CE‑SDS PLUS assays on the same instrument to increase throughput and standardize your protein size analysis across all stages of drug development.
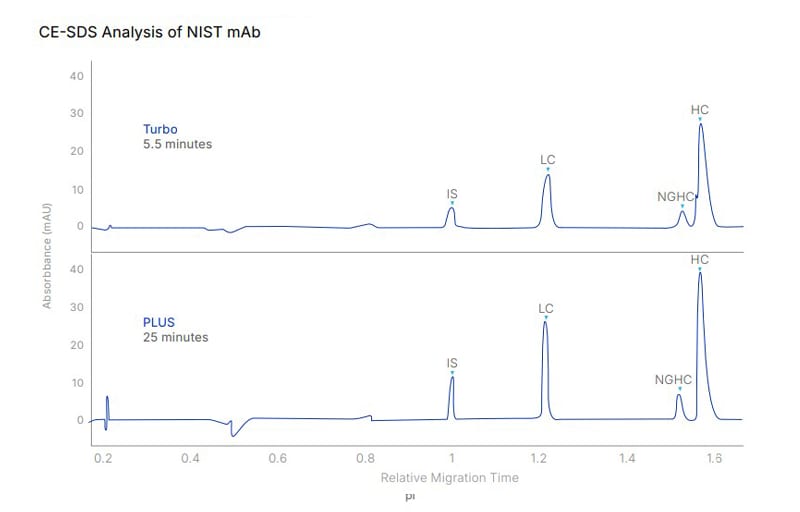
Figure 3: A comparison of size separation of NIST mAb obtained from the Maurice Turbo CE-SDS cartridge and the CE-SDS PLUS cartridge, run on the Maurice system. Both methods are comparable.
AAV Analysis for Gene Therapy Workflows
Analyze AAV CQAs with CE on a single instrument. Use Maurice to evaluate AAV identity, purity, capsid stability, and charge heterogeneity with the same platform you use for protein analytics. Consolidating these methods helps gene therapy teams rapidly assess critical quality attributes using reproducible CE‑based workflows.
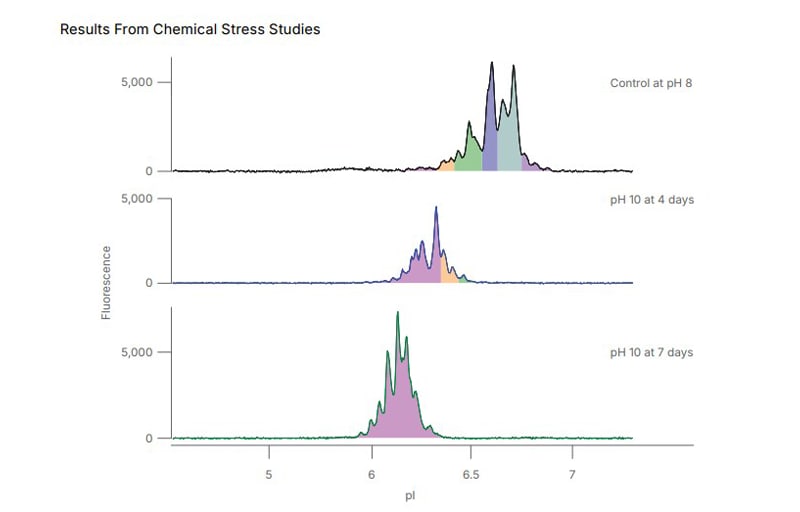
Figure 4: Stability of AAV8 under forced chemical stress monitored with icIEF, showing a clear increase in acidic species as the duration of stress increases.
Next-Gen Therapeutic Development
Advance next‑gen biologics with CE analytics. Support the development of ADCs, bispecifics, fusion proteins, and viral‑vector‑based therapies by combining CE‑SDS, icIEF, and icIEF fractionation into one automated system. Use Maurice and MauriceFlex to accelerate characterization, manage modality‑specific complexity, and maintain confidence in product quality across development.
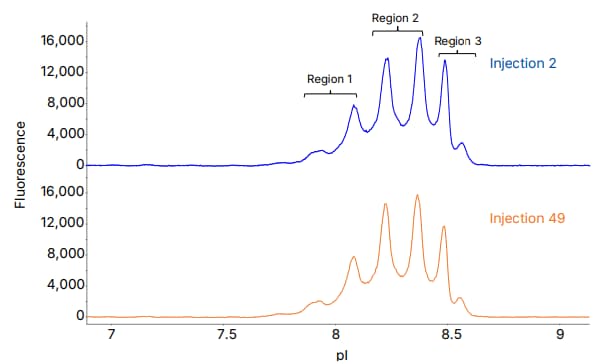
Figure 5: Analysis of an ADC using icIEF with Maurice OBM, showing reproducible results alongside preserving sample stability over a batch of 49 injections.
