Micro-Flow Imaging (MFI) Applications
The detection of subvisible particles is a critical step in biotherapeutic development to determine the safety of a biotherapeutic. Micro-Flow Imaging (MFI) detects different types of particles, from glass shards to protein aggregates as small as 1 µm. MFI is used for particle analysis in formulation development of different biotherapeutics.
Formulation Development | Quality Control | Gene Therapy | Biosimilar Development

MFI particle classifications based on conventions formalized by Pharmacopeial agencies. These morphological measurements help pharmaceutical companies and regulators assess the quality and safety of biopharmaceutical products.
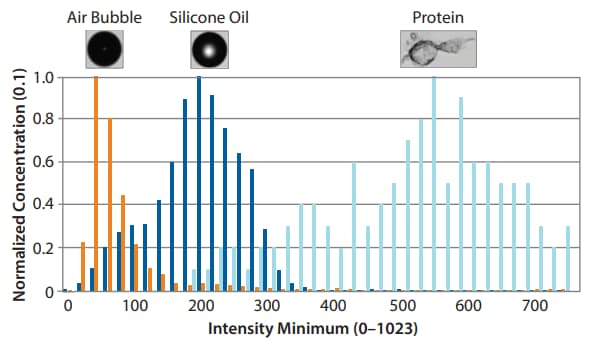
Differentiation of Protein Aggregates from Non-Aggregates. The MFI 5000 Series gives you the size, concentration and image for each particle in just one test and then lets you isolate them by different sub-populations.
Gene Therapy
Assess the stability of your AAVs under various stress conditions. Identify aggregates and impurities in your samples with higher sensitivity on MFI Instruments than light obscuration methods.
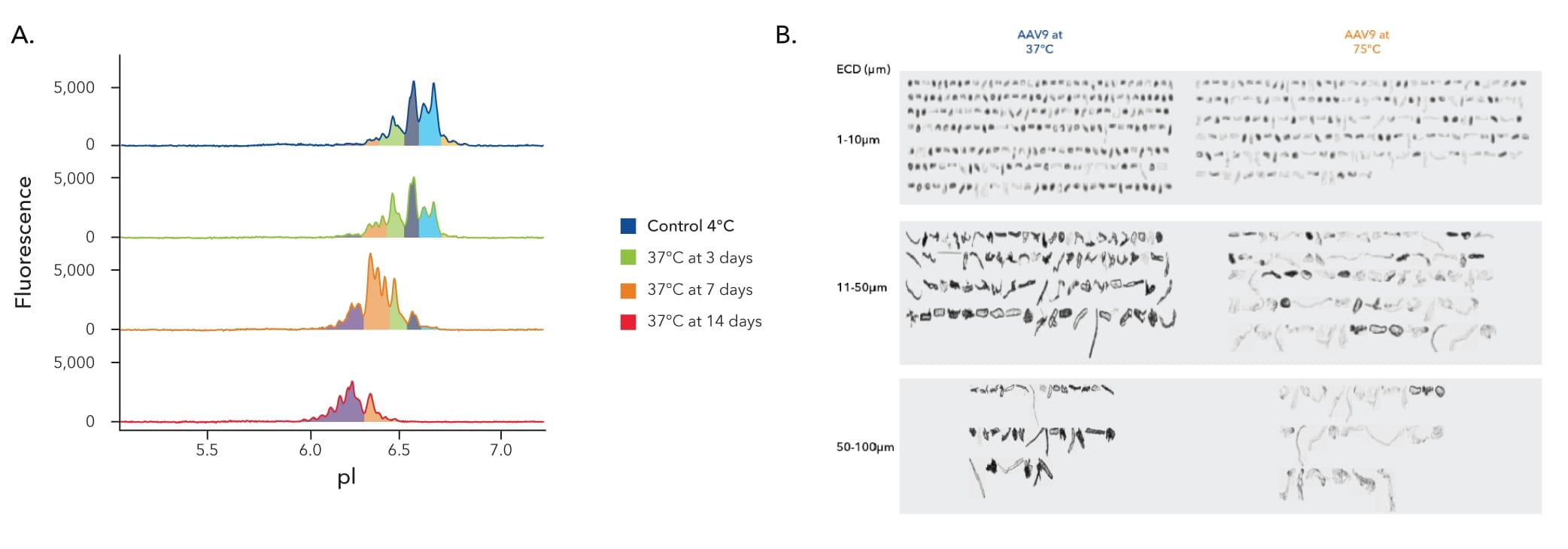
Stability assessment plays a pivotal role in ensuring AAV vector particles maintain their structural and functional integrity over time, minimizing impacts on potency, safety, and efficacy. Typical methods of AAV stability analysis, such as dynamic light scattering (DLS) and size exclusion chromatography (SEC) are limited by resolution and offer varying degrees of quantification.
By moving to a direct detection method, you can get higher levels of sensitivity in less time, providing robust stability data to simplify regulatory approvals.
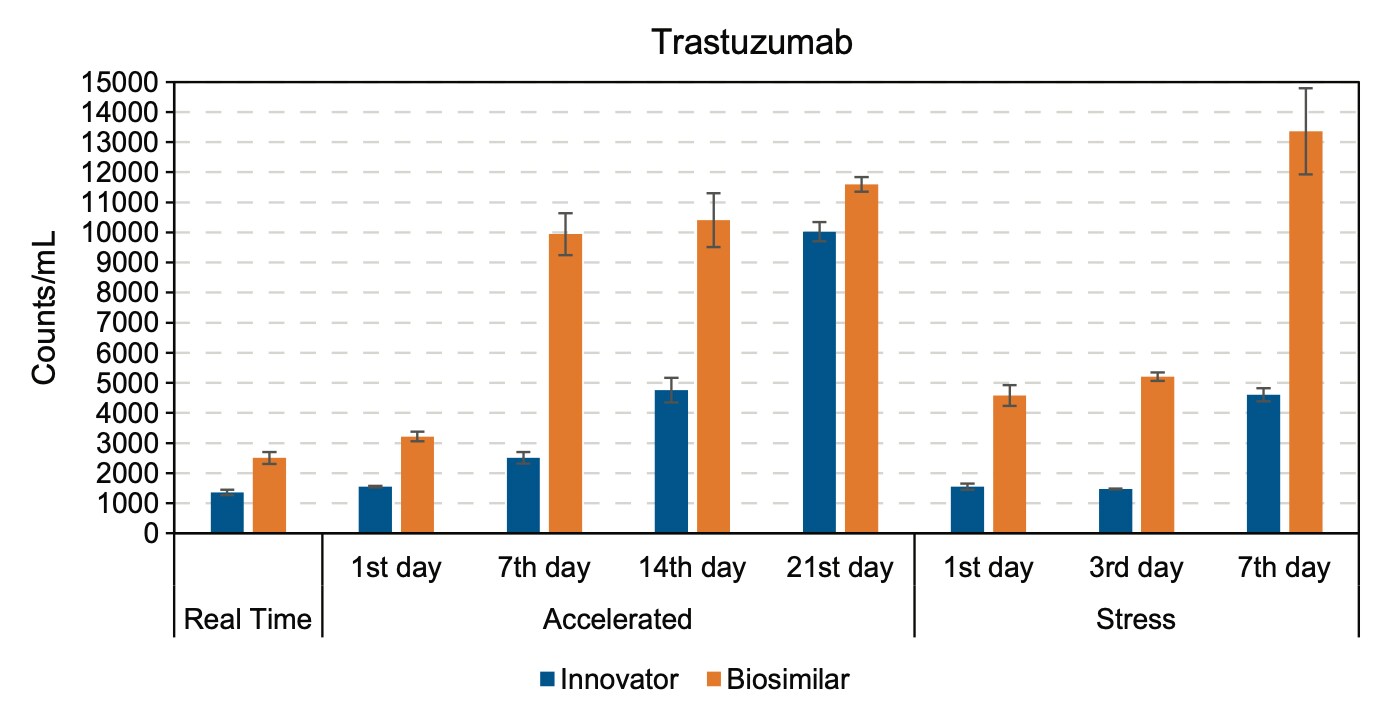
Particle formation of trastuzumab innovator and biosimilar during real time, accelerated, and stress tests. Shown are mean values with error bars representing the standard deviations of two replicates.
