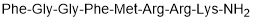Having trouble trying to find the right product? Chat with our scientists below - we're here to help!
326 results for "CXCR7/RDC-1" in 产品
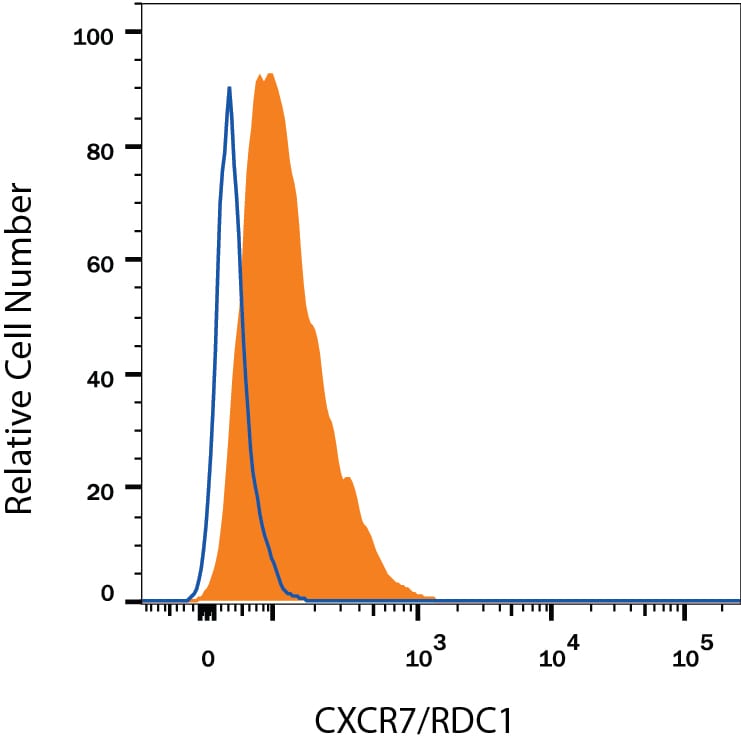
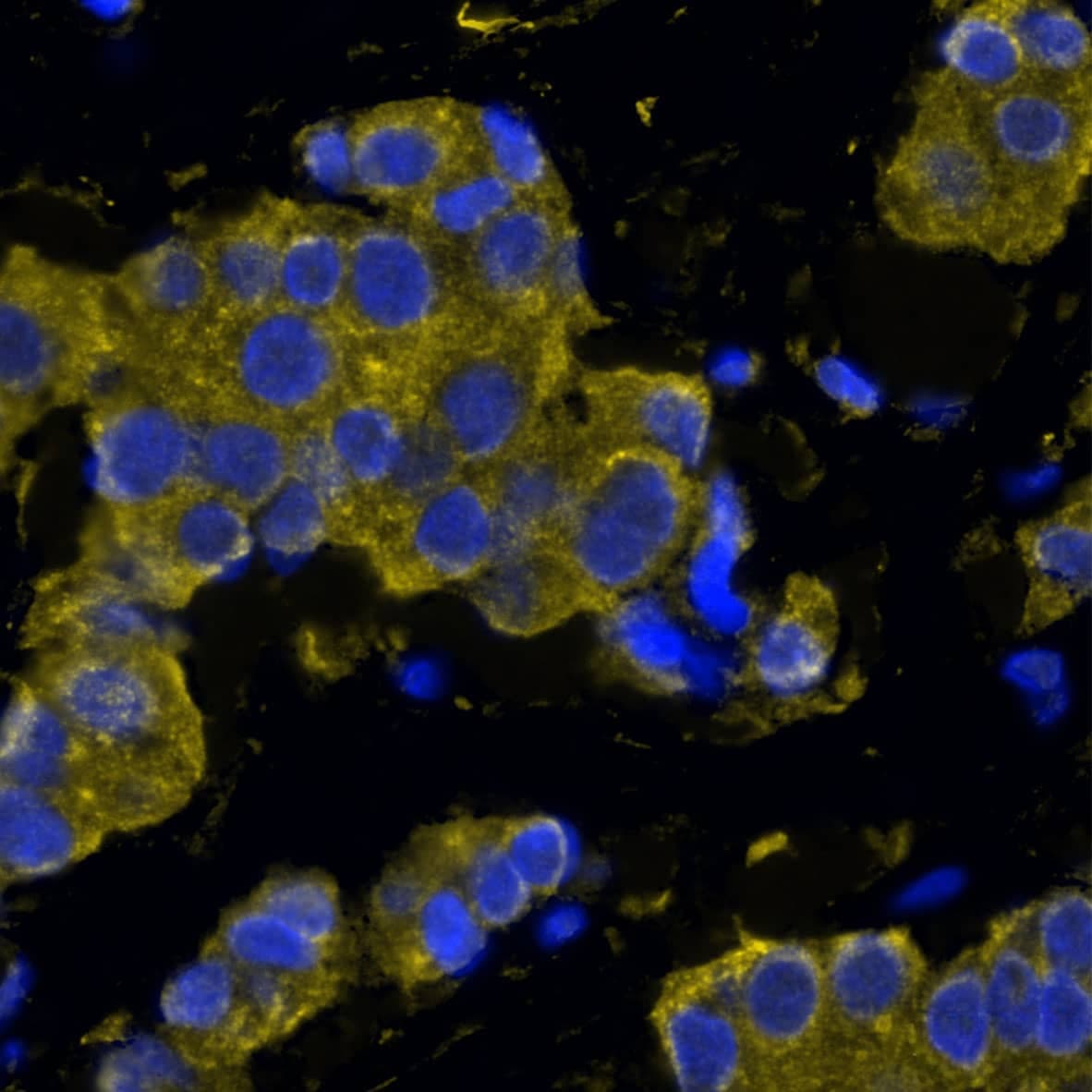
CXCR7/RDC-1 Products
CXCR7/RDC-1 is a G protein-coupled receptor (GPCR) member of the CXC subfamily of chemokine receptors. Human CXCR7/RDC-1 is 362 amino acids (aa) in length with a predicted molecular weight of 41 kDa. Mouse and rat CXCR7/RDC-1 share 93% aa sequence identity with the human protein. CXCR7/RDC-1 binds to and acts as a scavenger for CXCL11/I-TAC and CXCL12/SDF-1. CXCR7/RDC-1 can also function as a co-receptor for HIV and SIV. Unlike other chemokine receptors, CXCR7/RDC-1 does not activate G protein ...
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #11G8 |
| Applications: | IHC, Flow, CyTOF-ready |
Loading...
| Reactivity: | Human, Mouse, Rat, Sheep |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB, ICC/IF, Simple Western, Flow (-) |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG2A Monoclonal Clone #358426 |
| Applications: | mIF, IHC, Flow, COMET, CyTOF-ready |
Loading...
| Reactivity: | Human, Mouse, Rat |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB, ELISA, Flow, ICC/IF, + 1 More |
Loading...
| Reactivity: | Mouse |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB, Flow, CyTOF-ready |
Loading...
| Reactivity: | Human, Mouse |
| Details: | Mouse IgG1 Monoclonal Clone #11G8 |
| Applications: | Flow |
Loading...
| Reactivity: | Mouse |
| Details: | Rat IgM Monoclonal Clone #734110 |
| Applications: | Flow, CyTOF-ready |
Loading...
Recombinant Monoclonal Antibody.
| Reactivity: | Human |
| Details: | Mouse IgG2A Monoclonal Clone #358426R |
| Applications: | IHC, Flow, CyTOF-ready |
Loading...
Recombinant Monoclonal Antibody.
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #11G8R |
| Applications: | IHC, Flow, CyTOF-ready |
Loading...
| Reactivity: | Human, Mouse |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB, ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG2A Monoclonal Clone #358440 |
| Applications: | IHC, ICC/IF |
Loading...
| Reactivity: | Human, Mouse |
| Details: | Mouse IgG1 Monoclonal Clone #11G8 |
| Applications: | Flow |
Loading...
| Reactivity: | Rat |
| Details: | Mouse IgG2A Monoclonal Clone #896032 |
| Applications: | Flow, CyTOF-ready |
Loading...
Recombinant Monoclonal Antibody.
| Reactivity: | Rat |
| Details: | Mouse IgG2A Monoclonal Clone #896032R |
| Applications: | Flow, CyTOF-ready |
Loading...
| Reactivity: | Human |
Loading...
| Reactivity: | Mouse |
Loading...
| Reactivity: | Rat |
Loading...
| Reactivity: | Human |
Loading...
Atypical chemokine receptor (ACKR3) agonist
| Purity: | ≥95% |
Loading...
| Applications: | AC |
Loading...
| Reactivity: | Mouse |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB, Flow, CyTOF-ready |
Loading...
| Reactivity: | Mouse |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB, Flow, CyTOF-ready |
Loading...
| Reactivity: | Mouse |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB, Flow, CyTOF-ready |
Loading...
| Reactivity: | Mouse |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB, Flow, CyTOF-ready |
Loading...
| Reactivity: | Mouse |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB, Flow, CyTOF-ready |
Loading...



![Simple Western: CXCR7/RDC-1 AntibodyBSA Free [NBP2-24779] Simple Western: CXCR7/RDC-1 AntibodyBSA Free [NBP2-24779]](https://resources.rndsystems.com/images/products/CXCR7-RDC-1-Antibody-Simple-Western-NBP2-24779-img0003.jpg)
![Western Blot: CXCR7/RDC-1 Antibody [NBP1-31309] Western Blot: CXCR7/RDC-1 Antibody [NBP1-31309]](https://resources.rndsystems.com/images/products/CXCR7-RDC-1-Antibody-Western-Blot-NBP1-31309-img0012.jpg)
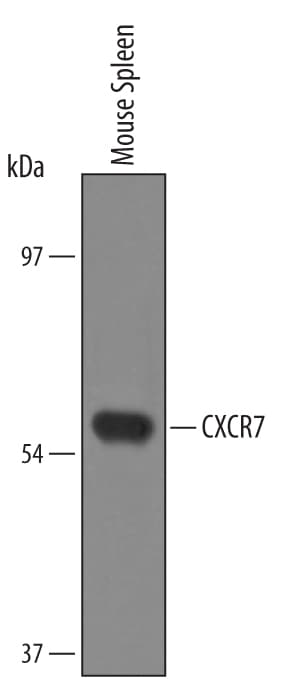
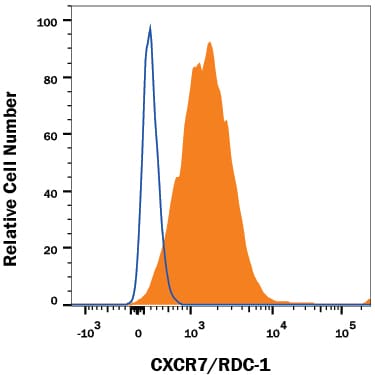
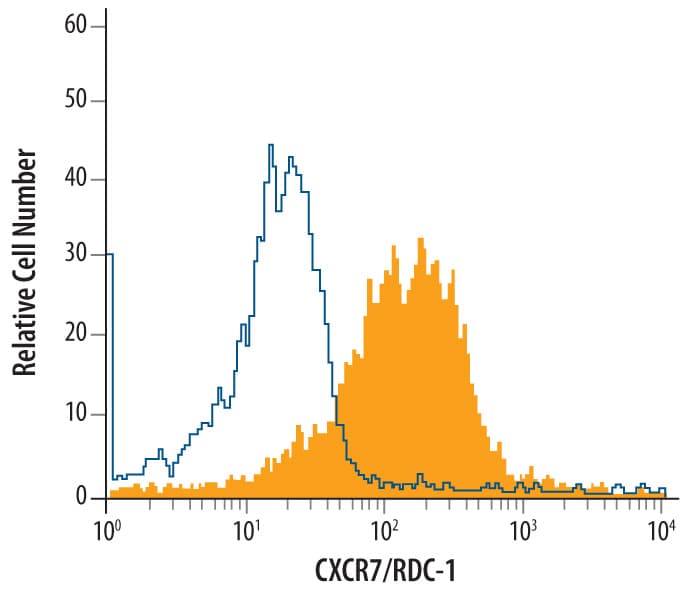
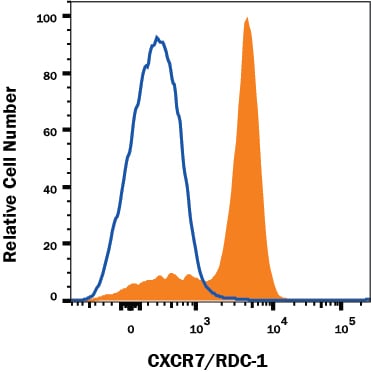
![Western Blot: CXCR7/RDC-1 Antibody [NBP2-58162] Western Blot: CXCR7/RDC-1 Antibody [NBP2-58162]](https://resources.rndsystems.com/images/products/CXCR7-RDC-1-Antibody-Western-Blot-NBP2-58162-img0002.jpg)